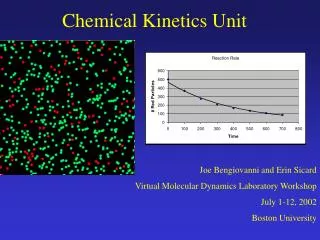
Chemical Kinetics Unit
Chemical Kinetics Unit. Joe Bengiovanni and Erin Sicard Virtual Molecular Dynamics Laboratory Workshop July 1-12, 2002 Boston University. Introduction. Topics covered: collision theory, activation energy, catalysis, rates of reactions, first-order reactions, second-order reactions.

Chemical Kinetics Unit
E N D
Presentation Transcript
Chemical Kinetics Unit Joe Bengiovanni and Erin Sicard Virtual Molecular Dynamics Laboratory Workshop July 1-12, 2002 Boston University
Introduction Topics covered: collision theory, activation energy, catalysis, rates of reactions, first-order reactions, second-order reactions • Unit provides a logical progression through a difficult topic in Chemistry. • Wet lab components • Computer simulation components • -serve to enhance learning and understanding of the material. • -give students concrete data they can work with. • Students will inquire about phenomenon observed in demonstrations and Simulabs.
Audience • Designed for 11th grade college bound chemistry I students • Students who struggle with conceptual/abstract ideas will benefit from simulations • MCAS requirement: -students will have to pass 2 science tests to graduate • -comprehensive understanding is required to pass this test • SAT II: 7% of test questions are from equilibrium and reaction rates topics
Placement in the Curriculum • Unit adheres to Massachusetts State Frameworks • Unit directly follows unit on solutions • Students will already have insight on dynamic nature of chemical reactions • -Gas laws, thermochemistry, reactions . . . . • Simulabs will be used to enhance learning in addition to wet labs
Goals • Students gain understanding of the dynamic nature of reactions • Students apply conceptual knowledge to practical activities • Students understand that “Chemical reactions occur at different rates.” • –Massachusetts State Frameworks
Student Objectives • Students will be able to: • Discuss the dependence of reaction rates on concentration, temperature, and surface • area • Describe the importance of molecular collisions to reaction rates • Define and show how to calculate activation energy • Explain how a catalyst/enzyme affects a reaction • Contrast first order and second order reactions • Determine reaction rates from experimental data • Practice techniques of data collection and graphical analysis
Time • Approximately six 50 minute class periods and two 90 minute lab periods • Major preparation: Crystal Violet lab, Clock lab • Students can help set up/take down some activities: Toothpickase, Buret lab • Preparation of demonstrations • Kinetics Unit directly followed by Equilibrium Unit
Computer Needs • 8-10 computers in classroom to accommodate groups of 2-3 students • A full computer lab (at least 24 computers) 1 per student • Projection or television with computer hookup to show Simulab demonstrations in • class • VMDL software and Simulab files
Resources Simulabs: VMDL software and Simulab files Crystal Violet lab: Vernier Colorimeter Calculator Based Laboratory (CBL) System Vernier Adaptor Cable Crystal violet chemical Graphing calculator Demonstrations: 30% Hydrogen Peroxide (try pool shock) Plaster
Unit Time Line Day 1 Discussion -Intro to reactions. Discussion -Collision theory, rates in general (slow, fast, medium). . Demonstrations -rust, Mg +HCl, Plaster Day 2 Discussion -Factors affecting reaction rates (temp, pressure, volume, concentration, surface area) Demonstration -Candle/Candle-1 Simulab Assignment -Design an Experiment project
Unit Time Line Day 3 -Double lab period Discussion -Activation energy Demonstration -MnO2 + H2O2 Activity -Toothpickase Discussion -Intro to catalyst and enzymes Activity -The Clock Reaction Assignment -The Clock Reaction –Graphical analysis Day 4 Discussion -Rate laws Activity -1st order Simulab Assignment -Answer Simulab questions homework Day 5 Review -1st order Simulab- hand in results and graphs Activity -Kinetics of a Draining Buret mini wet lab Classwork -Buret lab questions 1-8
Unit Time Line Day 6 Review -Day 5 classwork Discussion -2nd order reactions Demonstration -2nd order Simulab Day 7 –Double Lab periodLab Activity -Rate law determination of the Crystal Violet Reaction Assignment -Full lab report write up due 1 week from date of lab Day 8 Discussion -Review Crystal Violet Lab Quiz -Rate laws Click here for outline text file
Instructional Activities “Toothpickase” -Illustrates the relationship between concentration and rate -Demonstrates how enzymes are catalysts Kinetics of a -Practice in determining reaction rates Draining Buret First Order Simulab -Provides a dynamic picture of reactions for visual learners Rate Law Determination -Students use experimental conditions to support study of of the Crystal Violet reaction rates Reaction The Clock Reaction –Examines important factors (concentration and temperature) that affect reaction rates
Demonstrations Rust Plaster Mg+HCl MnO2 + H2O2 -show examples of rusted metal and non-rusted metal (slow reaction) -mix a bit of plaster with water at beginning of class. By end of class it should stiffen and get warmer. (medium rate, exothermic reaction) -mix a bit of Mg(s) with dilute HCl and see the immediate evolution of gas. (fast reaction) Collect gas in an inverted test tube and ignite with a glowing splint for added effect. -Using 3% H2O2, add a few grams of MnO2 to show catalyzed decomposition of H2O2 to H2O and ½ O2. Ignite a glowing splint over gas to show oxygen production. For added effect, add a drop of food coloring to 30% H2O2 in a large graduated cylinder then add MnO2.
Simulab Demonstrations Candle/Candle-1 Simulab -Show these 2 simulations side by side in order to demonstrate the importance of surface area on reaction rates. Candle Simulations are found under Universal/Additional Files. 2nd Order Simulab -This can be done as a whole class activity. Follow procedure 2.2 Second order reaction from page 13 of the yellow book. Data tables can be constructed on the board by the teacher and volunteers and each student can sketch the graphs from the data. Please note: Any of the Simulabs can be used for demonstration purposes if time or resources are limiting.
Adjustments/Adaptations • Reac2000 Simulab may replace Clock lab part II: Effects of Temperature • If computers are not easily available, only one Simulab may be done • - For instance, do only the first order of reactions simulab • - Adapt the others as in-class demos and discussions • Have a single teacher-run Simulab and do the analysis as a class. • Supplement catalyst information with addition of Catalyst Simulab
Assessments • First order Simulab questions and graphs from Yellow Book page 10 • Toothpickase graph and questions homework • Experimental Design performance assessment • Draining Buret activity question • Crystal Violet lab report -rubric • Lab writeup section of Buret Kinetics lab may also be assigned (optional)
References Toothpickase Activity http://sps.k12.ar.us/massengale/toothpickase.htm SAT II Chemistry Subject Test Format- The College Board http://www.collegeboard.com/sat/center2/chem/format.html Massachusetts State Frameworks- Department of Education http://www.doe.mass.edu/frameworks/current.html Vernier Software and Technology http://www.vernier.com/index.html Addison-Wesley, Chemistry Laboratory Manual, Prentice Hall 2000 Virtual Molecular Dynamics Laboratory Manuals and Software, Center for Polymer Studies, Boston University 2002
Contact Info Joe Bengiovanni - Bengi71@yahoo.com Erin Sicard - Erinsicard@yahoo.com