Tests for Positive Ions.
130 likes | 163 Views
Learn to identify various positive ions through qualitative tests. Understand procedures and observations for different ions. Safely conduct experiments to identify known and unknown substances. Enhance your chemistry skills in ion analysis.

Tests for Positive Ions.
E N D
Presentation Transcript
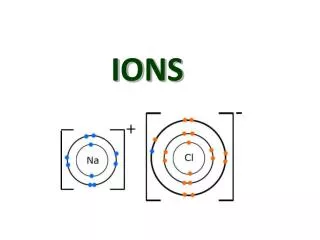
Tests for Positive Ions. 2.) Qualitative tests.
Learning Objectives. By the end of the session: • Everyone will have experience of carrying out qualitative tests for positive ions. • Most students will be able to identify Aluminium, Calcium, Copper (II), Iron (II), Iron (III) Ammonium and Magnesium ions. • A few students will be able to identify an unknown ion using qualitative tests.
Starter. • We have seen that flame tests allow us to identify some metal ions. • If I try to carry out a flame test on a compound containing Aluminium, Magnesium or Lead, what do you notice? What did we notice about the flame colours of Calcium and Lithium? • Some elements have flame colours which are very similar. This makes them difficult to identify. • Today, we will look at another technique to help us differentiate between compounds which give similar flame tests.
Safety. • Sodium hydroxide is corrosive. • Treat all known and unknown ions as if they were toxic. • Ammonia gas is harmful and should not be inhaled. • Care needed when heating Sodium hydroxide solution.
Qualitative tests. Using the apparatus on the previous slide, this is the procedure for qualitative tests. • Place approximately 1cm3 of the substance under test, in a test tube. • Add 1cm3 of dilute Sodium Hydroxide solution. • Carefully observe. Note your observations. • Repeat steps 2 & 3 twice more.
Observations. • You should now have a complete set of observations for the test-tube reactions. • Repeat the test for Ammonium ions, but this time, gently heat the test-tube over a Bunsen burner. Hold a piece of damp red litmus paper to the mouth of the test-tube.What do you see? What does this mean? Why moisten the litmus paper?
Unknown substance. • Test the unknown substance with Sodium Hydroxide solution. Note your observations and compare with the results of your experiments. Can you identify the unknown? • X =
Unknown substance. • Test the unknown substance with Sodium Hydroxide solution. Note your observations and compare with the results of your experiments. Can you identify the unknown? • X =
Homework. Use the following observations to identify the unknown ions.
Learning Objectives. By the end of the session: • Everyone will have experience of carrying out qualitative tests for positive ions. • Most students will be able to identify Aluminium, Calcium, Copper (II), Iron (II), Iron (III) Ammonium and Magnesium ions. • A few students will be able to identify an unknown ion using qualitative tests.