Kwl chart--moles
Kwl chart--moles. MOLES. This unit test contains 6 types of problems: Molar mass and % composition—must be able to write a chemical formula Grams to moles (using molar mass) Moles to particles/atoms/molecules (using Avogadro’s #)

Kwl chart--moles
E N D
Presentation Transcript
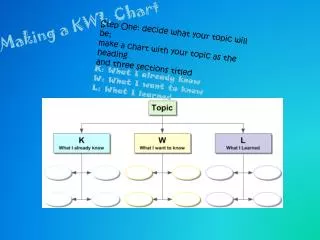
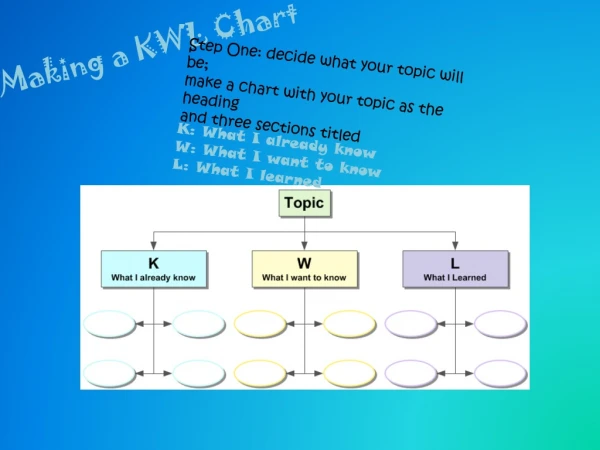
MOLES • This unit test contains 6 types of problems: • Molar mass and % composition—must be able to write a chemical formula • Grams to moles (using molar mass) • Moles to particles/atoms/molecules (using Avogadro’s #) • Grams to moles to particles (using molar mass and Avogadro’s #) • Empirical formulas • Molecular formulas
Mole • The SI (metric) unit used to measure the amount of a substance • 1 mole is always equal to: --Molar mass (g/mole) --Avogadro’s number of particles (6.02 x 1023) --22. 4 Liters of a GAS (AKA molar volume) These may be used as conversion factors when working mole problems.
activator • Define molar mass AND Avogadro’s number.
Molar mass of elements—same as atomic mass • Ex: Titanium 47.867 = 47.9 g/mole (this sample contains Avogadro’s number of atoms) • Ex: oxygen 15.999 = 16.0 g/mole (this sample contains Avogadro’s number of atoms)
Molar Mass of Compounds—must write chemical formula correctly • Multiply the # of atoms for each element by the atomic mass from periodic table Ex:Magnesium hydroxide Mg (OH)2 Mg 1(24.3) = 24.3 O 2(16.0) = 32.0 H 2(1.0) = 2.0 58.3 g/mole (this mass also contains Avogadro’s number of molecules)
Find the molar mass of aluminum sulfate Al 2 (SO4)3 Al 2(27.0) = 54.0 S 3 (32.1)= 96.3 O 12(16.0) = 192.0 342.3 g/mole (this mass also contains Avogadro’s number of molecules)
MOLAR MASS PRACTICE-- • Stannic carbonate • Diarsenicpentasulfide • Hydrofluoric acid
ACTIVATOR: calculate the molar mass • Sucrose
% composition • Shows the % of each element that makes up a compound • Must be calculate molar mass first. Ex: magnesium hydroxide Mg (OH)2 Mg 1 x 24.3 =24.3 24.3/58.3 x 100 = 41.7% O 2 x 16= 32.0 32.0/58.3 x 100 = 54.9% H 2x1.0 = 2.0 58. 3 g/mole 2.0/58.3 x 100 = 3.4%
ACTIVATOR: • Calculate the % composition of sulfurous acid
LAB—BITE THE BUBBLE • SAVE YOUR WRAPPER FOR ENTIRE LAB!! • DO NOT START CHEWING UNTIL YOU SIT DOWN BACK AT YOUR DESK. • CHECK BALANCE TO MAKE SURE IT’S OK BEFORE YOU START!!
READ PROBLEM: • MAKE A HYOTHESIS: • PROCEDURE 1—4 • DATA TABLE 1—5
After chewing: (KEEP YOUR SAME BALANCE) • Procedure 5—8 • Data table 6—8 • Conclusion • Questions 1—2
Calculate the % composition for a sugar substitute called SUCRALOSE
C 12( 12.0) = 144.0 • H 19 (1.0) = 19.0 • Cl 3 (35.5) = 106.5 • O 8 (16.0) = 128.0 397. 5 g/mole % C= 144.0 / 397.5 x 100 = 36.2% % H= 19.0 / 397.5 x 100 = 4.8% % Cl= 106.5 / 397.5 x 100 = 26.8% % O= 128.0/ 397.5 x 100 = 32.2%
Ticket out • 1. Calculate the % composition of carbonic acid. • 2. Calculate the % composition of diantimony trioxide.
1. H2CO3 H-2 (1.0) = 2.0 3.2% C- 1 (12.0) = 12.0 19.4% O – 3(16.0) = 48.0 77.4% 62.0 g/mole • Sb2O3 Sb- 2(121.8) =243.6 83.5% O – 3(16.0) = 48.0 16.5% 291.6 g/mole
ACTIVATOR • Calculate the molar mass AND % composition of: • C12H22O11 • Cupric sulfate
C: 12 (12.0) = 144.0 • H: 22 (1.0) = 22.0 • O: 11(16.0) = 176.0 CuSO4 Cu: 1(63.5)= 63.5 S: 1 (32.1) = 32.1 O: 4(16.0) = 64.0
Converting Grams to moles Will need to use unit conversion(cancellation) and molar mass will be used for the conversion factor. Ex: 2.50 grams of hydrochloric acid = ____moles H Cl 2.50 grams x 1 mole = 0.0685 moles(3sigfigs) 36.5 grams
Converting moles to grams Ex: 2.50 moles of HCl = __________grams 2.5 moles x 36.5 grams = 91 grams 1 mole (2 sig figs)
Converting particles to moles • Particles, atoms, molecules (synonyms) • Will have to use Avogadro’s number as a conversion factor • Ex: 5.25 x 1025 atoms of Mg = _____moles 5. 25 x 1025 atoms x 1 mole = 87.2 moles 6.02 x 1023 (3 sig figs)
Converting moles to particles 2.50 moles MgO = _________molecules 2.50 moles x 6.02 x 1023 molecules 1 mole = 1.50 x 1024 molecules (3 sig figs)
Converting grams to particles • Will need to use both molar mass AND Avogadro’s number as conversion factors • Will be 2 steps instead of 1 step unit cancellation • Ex: 4.5 grams nitrous acid = __________molecules • HNO2 4.5 g x 1 mole x 6.02 x 1023 molecules = 47 g 1 mole 5.8 x 1022 molecules (2 sig figs)
Converting particles to grams • Ex: 9.35 x 1021 particles of carbon tetrabromide = _____grams C Br4 9.35 x 1021 p x 1 mole x 154 grams = 6.02x1023 p 1 mole 2.39 grams (3 sig figs)
Answers to homework: must show work to receive credit 1. 0.14 mole (gram to moles) 2. 150 g (moles to grams) 3. 1.1 x 1023 molecules (g to molecules) 4 5.30 x 1025 molecules (moles to molecules) 5. 0.074 mole( gram to moles) 6. 0.619 g (particles to grams) 7. 0.49 mole (grams to moles) 8. 0.00083 mole (particles to moles)
Tab 3 “even more mole problems”—show work to receive credit • 22 g • 1.53 x 1024 molecules • 0.014 g • 7.2 x 1021 molecules • 7.2 x 1023 molecules • 2.08 x 106 g • 56 g • 2.5 g • 31 g • 0.0029 mole • 167 g BONUS: 3.37 x 1026 atoms
activator If grams are converted to moles, use _______________ to convert. If moles are converted to molecules, then use ______________to convert. ****Have calculator, periodic table, and best friend chart****
Mole game All members of your group must show their work on separate sheet of paper. When you calculate the answer, flip the card over to find a word. All of your words will make a sentence. First group to show all work and finish first, wins bonus!
Mole problem diagram Grams-----moles-----particles(atoms or molecules)
Extra practice-mole problems • 55.33 grams of sodium oxide = ____moles • 5.00 x 1022 particles of sodium= _______moles • 2.49 x 1026 atoms of acetic acid = _______________grams
Homework: Video sheet Problems #1—3, 5, 6
ACTIVATOR: Fill in the blanks with multiply/divide OR molar mass/Avogadro’s number: ***When going from moles to grams, _______________ by _____________. ***When going from moles to particles, ____________by _________________. I AM CHECKING 5 HOMEWORK PROBLEMS!!!!
Ticket out Briefly describe the steps for calculating an empirical formula AND molecular formula.
activator Define empirical formula ***both labs due today***
Mole review problems • Convert 5.03 x 1024 molecules of phosphoric acid to grams. • Convert 35.75 grams of dinitrogen monoxide to moles. • Convert 5.0 moles of water to molecules.
answers • 819 grams of H3PO4 • 0.8125 moles of N20 • 3.0 x 1024 molecules of water
Ticket out Tell how to solve for each: • G to moles • Moles to G • Particles to moles • Moles to particles • G to particles • Particles to G
Gum lab—show work to receive credit • HYPOTHESIS, DATA 1—8, CONCLUSION, QUESTIONS 1—4 • 3. MASS OF SUGAR (in grams—data #8)--------MOLES (SUGAR = C12 H22 O11) • 4. MOLES----------PARTICLES
Lab-using the mole • Data Table: mass of empty vial AND substances mass (make sure you’ve subtracted empty vial each time!!) • SHOW WORK TO GET CREDIT • CONVERT GRAMS----------MOLES • CONVERT MOLES---------PARTICLES • ANSWER QUESTIONS 1---5, 6 (BONUS)
Activator: Convert 25.0 moles of water to grams.
activator A molecular formula is a whole number____________of the empirical formula.
EMPIRICAL FORMULAS • Shows the SIMPLEST, WHOLE NUMBER ratio of elements in a compound • Will give you % composition of compound and ask you to find the formulas
Steps for calculating: 1. Change % sign to grams (some problems may already give you grams instead of %) 2. Convert grams to moles (using molar mass) **round to 4 decimals*** 3. Simplify the mole ratio by dividing each one by the smallest 4. Round to the nearest whole number and assign these numbers to the appropriate element
Example: A compound is 78.1% Boron and 21.9% H. Calculate the empirical formula. 78.1 grams B x 1 mole = 7.2315 moles B 10.8 g 21.9 grams H x 1mole =21.9 moles H 1.0 g 7.2315 : 21.9 7.2315 7.2315 1: 3 = BH3
Molecular formulas: Is a WHOLE NUMBER MULTIPLE of the empirical formula You must then first know the empirical formula