Practice Energy Calculation Quiz
E N D
Presentation Transcript
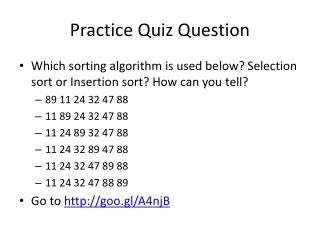
How much energy does it take to convert 722 grams of ice at 211C to steam at 675C?(Be sure to draw and label the appropriate heating or cooling curve.)Provided information:heat of fusion = 6.0 kJ/molheat of vaporization = 40.7 kJ/molspecific heat capacity of ice = 2.1 J/g∙Cspecific heat capacity of steam = 1.8 J/g∙C
Step 1: Convert the mass in grams to moles. 722 mole H2O 1 g H2O = 40.1 molH2O 18.0 g H2O
Step 2Heat the ice from 211C to its melting point of 0C. q = mcΔT q = (722 g)(2.1 J/g∙C)(0 (211C)) q = 3.20 x 105 J
Step 3Convert ice to liquid water - (melt the ice!!) q = ΔHfusion ∙moles q = (6.0 kJ/mol)(40.1 mol) q = 241 kJ = 241,000 J
Step 4Heat the ice from 0C to its boiling point of 100C. q = mcΔT q = (722 g)(4.18 J/g∙C)(100 0C) q = 302,000 J
Step 5Convert water to steam - (boil the water!!) q = ΔHvaporization ∙moles q = (40.7 kJ/mol)(40.1 mol) q = 1630 kJ = 1,630,000 J
Step 6Heat the steam from 100C to 675C. q = mcΔT q = (722 g)(1.8 J/g∙C)(675 100C) q = 747,000J
qtotal = q2+ q3 +q4 +q5+ q6 Step 7: Add the heats! qtotal= 3.2 x 105 J + 241, 000 J + 302,000 J + 1, 630,000 J + 747,000 J qtotal= 3,240,000 J or 3.24 x 106 J qtotal = 3,240 kJ or 3.24 x 103 kJ