The Enigmatic Ti3+ in Solar Nebula Object Inventory
Explore Ti valence shifts in chondrites and forsterite, revealing metamorphic insights. X-ray spectroscopy uncovers trivalent Ti, aiding nebular gas understanding.

The Enigmatic Ti3+ in Solar Nebula Object Inventory
E N D
Presentation Transcript
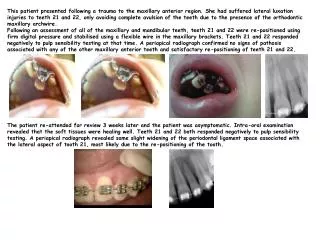
The Growing Inventory of Ti3+-bearing Objects from the Solar Nebula Steven B. Simon1, Stephen R. Sutton1, 2and Lawrence Grossman1, 3 1Dept. of the Geophysical Sciences, 2Center for Advanced Radiation Sources (CARS), 3The Enrico Fermi Institute The University of Chicago, Chicago, IL 60637 • Results:L3 – L6 chondrites • Some observations [9, 10] suggest that ordinary chondrites become more reduced from grades 3.0 to 4 and oxidized from grades 4 to 5 while other work indicates near-constant ƒO2s [11], leading to some uncertainty about the oxidation states and temperatures of ordinary chondrites during metamorphism [12]. We are investigating suites of L and LL chondrites to see how Ti valence varies with metamorphic grade, possibly providing a new constraint on conditions of chondrite metamorphism. Results for L chondrites are summarized in Fig. 5. More data are needed, but so far we have found that the average valence of Ti in olivine decreases from 4.0±0.1 to 3.7±0.1 with increase in grade from 3.0 through 3.6, and it is within 1s of 4 in the L3.8, L4 and L6 samples. Most of the Ti4+ is in the tetrahedral site, especially in the higher-grade samples. Pyroxene valences are ~3.5 in grades 3.0 – 3.6 and range from 3.5 to 4 in the L4 and L6 samples. As found in previous work [e. g., 4] there is no correlation between Ti valence and FeO content in olivine or pyroxene. 100 µm Fig. 2a En5 En6 En4 OL3 En7 En3 OL1 Meso1-4 En11 En1 En10 meso5 • Results:Semarkonachondrules • Type I (FeO-poor olivine and pyroxene) and Type II (FeO-rich) chondrules in Semarkona (LL3.0) were analyzed by XANES to see if they contain trivalent Ti [4]. This could help us determine whether the two types derive from different precursors or different crystallization environments. Results are shown in Fig. 2 below. Olivine in both chondrules shown contains Ti3+ but in different proportions and with different spatial distributions. Grain OL8 in the Type I chondrule 18 (red arrow in Fig. 2a) is unzoned in Fa but zoned with increasing Ti valence from ~3.0 in the core to ~3.6 at the rim (Fig. 2c). Grain OL5 (red arrow in Fig. 2b) in Type II chondrule 8 is strongly zoned in Fa but not in Ti valence (Fig. 2d). In chondrule 18 there is a strong inverse correlation between grain size and Ti valence in the cores of crystals (Fig. 2e). The systematics observed in the data for this chondrule are precisely what would be expected for oxidation during crystallization. The Type II chondrules contain Ti3+ but generally in lower proportions than the Type Is. The presence of Ti3+ in Type II chondrules suggests that their precursors were reduced and that the valence of Ti is more resistant to resetting than that of Fe. Introduction It has been known for some time that the Ti-rich phases fassaite, hibonite and rhönite, found in coarse-grained refractory inclusions, contain trivalent Ti, an indicator of formation under very reducing conditions. The development of Ti-XANES (X-ray absorption near-edge structure) spectroscopy has allowed measurement of Ti valence to be performed on relatively Ti-poor phases such as spinel and olivine. These are important phases among early solar system materials, and the valence of Ti in them can tell us whether they formed in a reducing, nebular gas or under more oxidizing conditions. We have investigated a wide variety of materials for the occurrence of Ti3+. This report is a summary of those findings. We have also recently begun an investigation of the valence of Ti as a function of metamorphic grade in L and LL chondrites, and some preliminary results are reported. Methods Samples were selected for analysis after characterization by scanning electron microscope and electron microprobe. Titanium K XANES spectra were collected using the GSECARS X-ray microprobe in fluorescence mode, with a 3 µm X-ray beam. Results were determined following the methods of [1, 2] and are reported as values from 3 to 4, representing the average Ti valences of the analytical volume. En2 meso6 O4 Dusty meso7 OL8 Fig.2b 200 µm G1 A En9 meso2 Chondrule 18 Type IAB OL5 OL4 Semarkona USNM 1805-18 OL8 meso6 meso7 OL9 meso3 OL2 OL3 OL7 meso4 meso5 OL1 meso1 • Results:Refractory forsterite • Isolated refractory forsterite (RF) grains (RF has CaO>0.4 wt% and Al2O3>0.2 wt%) and RF-bearing chondrules from two CM chondrites were studied by [3]. At least 75% of the Ti is trivalent in RF, whether it is found as isolated grains or in chondrules; RF has higher Ti3+/Ti4+ ratios than fassaite from refractory inclusions [3]. Trivalent Ti is also found in “typical” olivine in chondrules, but in lower proportions than in RF [4]. Fig. 5 Discussion That RF might contain Ti3+ was suggested by [13], who calculated refractory lithophile element contents of a melt that would be in equilibrium with RF. They predicted enrichments of ~10-40xC for most elements, but Ti enrichments of 50-65xCI would have to be inferred if it were only present as Ti4+. Our measurements of Ti3+/Ti4+ in RF combined with an olivine/liquid D for Ti3+ of 0.355 interpolated from Ds for V3+ and Sc3+ [14] indicate a liquid with 28xCI, well within the range predicted by [13] for other refractory lithophiles, supporting their model for the formation of RF. The lack of Ti3+ in Fo-bearing refractory inclusions (FOBs) is not understood. One of the inclusions we studied is an isotopically fractionated (FUN) inclusion, a type thought to have undergone evaporation, so it may have been exposed to oxidizing conditions, but the other FOB we studied is not a FUN inclusion. The occurrence of Ti3+ in Type I and Type II chondrules suggests that both types had reduced precursors. The valence of Ti in chondrules does not reflect the near-IW conditions under which FeO-bearing olivine forms, and the Fa contents of chondrule olivine do not reflect the reducing conditions (ƒO2≤IW-3) that stabilize Ti3+ in chondrule-composition melts [15]. Either Fe attains redox equilibrium more rapidly than Ti, or the Fe/Mg ratio of olivine and pyroxene can increase by inward diffusion of Fe without disturbance of the Ti3+/Ti4+ ratio. Measurements on L chondrites show that variation in Ti valence in olivine and pyroxene persists to metamorphic grades at which Mg/Fe ratios are equilibrated. The high proportions of tetrahedral Ti4+ indicate formation under anhydrous conditions [16]. These results and the chondrule data show that the Ti valence of olivine and pyroxene is not easily disturbed. Type IIA Type IAB Fig. 2e Fig. 2d Fig. 2c Fig. 2. (a, b)Backscattered electron images of a Type IAB (a) and a Type IIA chondrule from the Semarkona LL3 chondrite, showing locations of XANES analysis spots. Brightness of a mineral is proportional to the average atomic number of the elements in it. (c, d) Plots of mole % fayalite (Fa) and Ti valence as a function of distance across single olivine grains in Semarkonachondrules. No correlation is observed between Fa content and Ti valence within either grain. (e) Plot of Ti valence in phenocryst cores as a function of grain size. Ti valence in AOA olivine • Results:Amoeboid olivine aggregates (AOAs) • This is a common type of inclusion found in many carbonaceous chondrite groups. They have long been interpreted as nebular condensates [5, 6] and the presence of Ti3+ in them would support that idea. Analyses of olivine in AOAs in two CV3 chondrites, Allende and Efremovka, show that AOAs do contain Ti3+. As in chondrule olivine, Ti valence is not correlated with Fa content. As shown in Fig. 3 (left), the Ti in the relatively Fa-poor Efremovka olivine has a higher valence (avg. 3.7±0.1) than that in the more fayalitic Allende olivine (avg. 3.5±0.1). The presence of Ti3+ in both chondrule and AOA olivine is consistent with a common source, or perhaps related precursor assemblages, for these objects [7]. Fig. 1 Fig. 1.Plot of normalized intensity of pre-edge peak vs. energy; these parameters vary systematically as a function of valence and coordination number. Mixing lines are calculated from endmember values shown and are used to calculate valences in samples. Fig. 1 shows that almost all of the Ti in the isolated RF grains (open squares) is trivalent, Ti in the chondrule RF (open circles) is 80-90% trivalent, and that in forsterite-bearing inclusions is tetravalent. • Conclusions • 1. The occurrence of Ti3+ is not restricted to refractory inclusions; it is also found in olivine-rich objects such as chondrules and AOAs. • 2. The proportions of trivalent Ti in refractory forsterite and the resulting crystal/liquid distribution coefficient for Ti yield calculated residual liquid compositions with Ti contents within the range of other lithophile element enrichments predicted by Pack et al. [13], supporting their model. • 3. Ordinary chondrites retain Ti3+ at least through grade 6. The valence of Ti is not as easily reset as that of Fe. • Results:Forsterite-bearing refractory inclusions • Two of these objects (TS45, CG14) were investigated as a possible source of RF grains because they contain abundant, coarse, Ca-rich forsterite. It was found, however, as shown in Fig. 1, that neither the olivine nor the pyroxene in these inclusions contains Ti3+. Fig. 3 • Results: Murchison inclusions • Refractory inclusions in CM2 carbonaceous chondrites, such as Murchison, are dominated by oxides, mainly spinel and hibonite, unlike inclusions in CV3 chondrites, which are silicate-rich. Results [8] for five typical Murchison samples are shown in Fig. 4. Hibonite, spinel and pyroxene all contain Ti3+, and there is a good correlation of average Ti valence in spinel with that of coexisting pyroxene or hibonite. Pyroxene in Murchison inclusions generally has much lower Ti, Sc and V contents than fassaite in coarse-grained refractory inclusions, suggesting different sources, but the Murchison pyroxene has Ti3+/Ti4+ ratios within the range of fassaite from CV3 inclusions. References [1] Simon S. B. et al. (2007) GCA, 71, 3098–3118. [2] Farges F. et al. (1997) Phys. Rev. B,56, 1809-1819. [3] Simon S. B. et al. (2007) LPS XXXVIII, Abstract #1892. [4] Simon S. B. et al. (2008) LPS XXXIX,Abst. #1352. [5] Grossman L. & Steele I. (1976) GCA 40, 149-155. [6] Krot A. et al. (2004) ChemiederErde64, 185-239. [7]Simon S. B. et al. (2010) M&PS 45, A189. [8] Simon S. et al. (2009) LPS XL,Abst. #1626.[9] McSween H. Y. & Labotka T. (1993) GCA 57, 1105-1114. [10]Menzies O. et al. (2005) M&PS 38,1023-1042. [11] Kessel R. et al. (2004) M&PS39, 1287-1305. [12] Huss G. et al. (2006) MESS II, 567-586. [13] Pack A. et al. (2005) GCA69, 3159-3182. [14] Kennedy A. K. et al. (1993) EPSL115, 177-195. [15] Simon S. B. et al. (2011) LPS XLII, Abst. #1271. [16] Berry A. et al. (2007) Chem. Geol.242, 176-186. • Acknowledgments • This work was supported by NASA Grant NNX08AE06G (LG). Portions of this work were conducted at GeoSoilEnviroCARS (Sector 13), Advanced Photon Source (APS), Argonne National Laboratory. GeoSoilEnviroCARS is supported by NSF (EAR-0622171) and DOE-Geosciences (DE-FG02-94-ER14466). Fig. 4