POLYSACCHARIDES
POLYSACCHARIDES . By SANGH PARTAP Department of Pharmacy Teerthanker Mahaveer University, Moradabad, U.P., INDIA. Definition.

POLYSACCHARIDES
E N D
Presentation Transcript
POLYSACCHARIDES By SANGH PARTAP Department of Pharmacy TeerthankerMahaveer University, Moradabad, U.P., INDIA
Definition • Polysaccharides are high molecular weight polymers build up by repeated condensation of polyhydroxyaldehydes/ polyhydroxyketones which are joined together by glycosidic linkages, and can be hydrolysed to a large no. of monosaccharides.
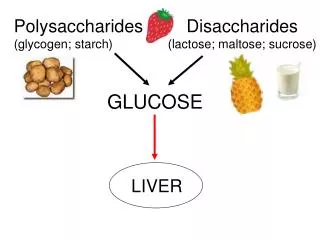
EXAMPLES ARE • CELLULOSE • INULIN • STARCH • CHITIN • HEPARIN • HYALURONIC ACID
Types of Polysaccharides • Homopolysaccharides :-also called as homoglycans. On hydrolysis yield only one type of monosaccharide unit. Example- cellulose, inulin, starch etc. • Heteropolysaccharides :- also called as heteroglycans. On hydrolysis yield more than one type of monosaccharide unit. Example- α- heparin, hyaluronic acid etc.
Cellulose • Cellulose is the chief constituent of the cell wall Of plant and most widely distributed carbohydrate. • Cellulose is an organic compound with the formula (C6H10O5)n, a polysaccharide consisting of a linear chain of several hundred to over ten thousand β(1→4) linked D-glucose units.
Types of cellulose • α- Cellulose :- is very much less degraded and does not dissolve in aqueous NaOH. • β- Cellulose:- is soluble in aqueous NaOH but insoluble in dill. Acid. • γ- Cellulose:- is insoluble in both.
Cellulose Structure and properties • Cellulose has no taste, is odorless, is hydrophilic, is insoluble in water and most organic solvents. Mol.formula of Cellulose has been found to be (C6H10O5)n from analytical data. When Cellulose is hydrolysed with fuming HCL it give D- glucose in 95-96% yield.This reaction reveal that Cellulose is made up of glucose unit.It means that the structure of Cellulose is based on the D- glucose unit.
Methylation,acetylation and nitration of Cellulose produce trisubstitution product as a max. substituted product, it means that each glucose unit present possesses 3 OH group in an uncombined state.
Fully methylated cellulose, when subjected to hydrolysis yield 2,3,6 tri-O-methyl D- glucose (90%) as main product and also 2,3,4,6 tetra-O-methyl D- glucose 0.6% as minor product.
Chemical method • Determination of the proportion of end group and comparing with the total no. of unit in molecule. Haworth methylation method:- Cellulose is completely methylated in inert atmosphere then methylated cellulose is hydrolysed by dill. Acid to cleave the glycosidiclinkage.thenonreducing end will yield 2,3,4,6 tetra-O-methyl D- glucose whereas all other will undergo hydrolysis to yield 2,3, tri-O-methyl D- glucose. The two hydrolytic product are seprated by chromatographic tech. Hence by knowing the % of tetra methyl derivative or ratio of tetra methyl- tri methyl derivative. This method is known as the end-group assay.
Periodic oxidation method • When cellulose is treated with sodium periodate or periodic acid,two mole of formic acids are obtained from reducing end whereas one mole of formic acid from the non-reducing end. the amount of formic acid is estimated by the titration method. This estimation gives the value of chain length.
Starch • Starch is a carbohydrate consisting of a large number of glucose units joined together by glycosidic bonds. This polysaccharide is produced by all green plants as an energy store. • It is the most important carbohydrate in the human diet and is contained in such staple foods as potatoes, wheat, maize (corn), rice, and cassava.
Pure starch is a white, tasteless and odorless powder that is insoluble in cold water or alcohol. • It is produced in the green plant by the reaction b/w water and oxygen under catalytic effect of chlorophyll and sunlight.
It consists of two types of molecules: • the linear and helicalamylose:-Amylose is a linear polymer made up of D-glucose units. • the branched amylopectin: Branching takes place with α(1→6) bonds occurring every 24 to 30 glucose units. • Depending on the plant, starch generally contains 20 to 25% amylose and 75 to 80% amylopectin.
Constitutions of amylose • It is empirical formula is C6H10O5 . • On complete hydrolysis amylose give D-glucose units .this indicate that amylose is composed of only D-glucose units.
Enzymatic hydrolysis amylose give maltose. Maltose is 4-o-(α-glucopyranosyl) –D-glucopyranose, all the glucose unit in starch are linked through C1α and C4. hence amyloseposesses the foolowing str. Which explain the hydrolysis product. • The amylose is confirmed by hydrolysis of its fully methylated derivative to 2,3,6 tri-o-methyl-D-glucopyranose and 0.32% of 2,3,4,6 tetra-o-methyl-D-glucopyranose
Constitution of amylopectin • Its empirical formula is C6H10O5 • On complete hydrolysisamylopectin give D-glucose units .this indicate that amylose is composed of only D-glucose units. • Fully methylatedamylopectin on hydrolysis gives 2,3,6 tri-o-methyl-d-glucose and 0.32% of 2,3,4,6 tetra-o-methyl-d-glucose.
Differences between starch & cellulose STARCH • It is reserve food material of plant and is found mainly in seeds, roots and tubers of the plant. Wheat , maize, patatoes and rice are its main commercial source. • It can be separated in two components, amylose and amylopectin. CELLULOSE • It is very widely distribution in nature as the chief component of wood and plant fibres. Cotton, wood and juite are its main source. • It is single compound which can not be separated into or more components.
STARCH CELLULOSE • Both amylose and amylopectin consists of D-(+) glucose unit linked through α-glucoside linkage. • It gives blue colour with iodine. • It is also made up of D-(+) glucose unbrached chain but they are interlinked by β-glucoside linkage. • It does not give blue colour with iodine.
CHITIN • Linear homopolysaccharide composed of N-acetyl glucosamine residues in linkage • The only chemical difference from cellulose is the replacement of the hydroxyl group at C-2 with an acetylated amino group. • Chitin forms extended fibers similar to those of cellulose, and like cellulose cannot be digested by vertebrates. • Chitin is the principal component of the hard exoskeletons of nearly a million species of arthropods—insects, lobsters, and crabs, for example— and is probably the second most abundant polysaccharide, next to cellulose, in nature.
Hetropolysaccharides • Hyaluronic acid • Heparin • Agarose Hyaluronic acid • Hyaluronic acid is an important GAG (Glycosamonoglycan) found in the ground substance of synovial fluid of joints and vitreous humor eye. • serves as a lubricant and shock absorbant in joints. • composed of alternate unit of D-gluconicacidN-acetyl D-glucosamine.
Heparin • Heparin is an anticoagulant( prevant blood clothing) produced by basophils and mast cells that occur in blood, lung, liver, kidney, spleen etc. • Heparin is composed of alternate unit of N-sulfo D-glucosamine 6-sulfate and glu 2-sulfate.
Agarose The repeating unit consists of D-galactose (β 1 4)-linked to 3,6-anhydro-L-galactose (in which an ether ring connects C-3 and C-6). These units are joined by ((α1 3)-) glycosidic links to form a polymer 600 to 700 residues long. A small fraction of the 3,6-anhydrogalactose residues have a sulfate ester at C-2.
Uses • Agarose gels are used as inert supports for the electro pho- retic separation of nucleic acids, an essential part of the DNA sequencing process . • Agar is also used to form a surface for the growth of bacterial colonies. • Another commercial use of agar is for the capsules in which some vitamins and drugs are packaged; the dried agar material dissolves readily in the stomach and is metaboli- -cally inert.
Polysaccharides (glycans) serve as stored fuel and as structural components of cell walls and extracellular matrix. • ■ The homopolysaccharides starch and glycogen are stored fuels in plant, animal, and bacterial cells. They consist of D-glucose with linkages, and all three contain some branches. • ■ The homopolysaccharides cellulose, chitin, and dextran serve structural roles. Cellulose, composed of (1n4)-linked D-glucose residues, lends strength and rigidity to plant cell walls. Chitin, a polymer of (1n4)-linked N-acetylglucosamine, strengthens the exoskeletons of arthropods. Dextran forms an • adhesive coat around certain bacteria. SUMMARY
Homopolysaccharides fold in three dimensions. The chair form of the pyranose ring is essentially rigid, so the conformation of the polymers is determined by rotation about the bonds to the oxygen on the anomeric carbon. • Starch and glycogen form helical structures with intrachain hydrogen bonding; cellulose and chitin form long, straight strands that interact with neighboring strands. • ■ Bacterial and algal cell walls are strengthened by hetero-polysaccharides—peptidoglycan in bacteria, agar in red algae. The repeating disaccharide in peptidoglycan is • GlcNAc(1n4)Mur2Ac; in agarose, it is D-Gal(1n4)3,6-anhydro-L-Gal.
Glycosaminoglycans are extracellular heteropolysacch- arides in which one of the two monosaccharide units is a uronic acid and the other an N-acetylated amino sugar. Sulfate esterson some of the hydroxyl groups give these polymers a high density of negative charge, forcing them to assume extended conformations. These polymers (hyaluronate, chondroitin sulfate, dermatan sulfate, keratan sulfate, and heparin) provide viscosity, adhesiveness, and tensile strength to the extracellular matrix.