Electromagnetic Spectrum Calculations
60 likes | 345 Views
Using Planck’s Constant…. Electromagnetic Spectrum Calculations. Who was Max Planck?. He was a German physicist and is considered as the founder of the quantum theory (Wikipedia 2011).

Electromagnetic Spectrum Calculations
E N D
Presentation Transcript
Using Planck’s Constant… Electromagnetic Spectrum Calculations
Who was Max Planck? • He was a German physicist and is considered as the founder of the quantum theory (Wikipedia 2011). • The symbol (h) is used to denote Planck’s constant, which he discovered in 1899. It is used as a proportionality constant between the energy and frequency of an electromagnetic wave (Wikipedia 2011). • h = 6.626 x 10-34 Js (joule seconds) Picture credit: http://en.wikipedia.org/wiki/File:Max_Planck.png
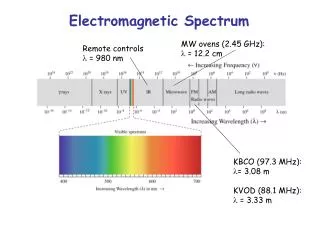
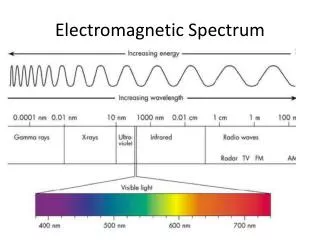
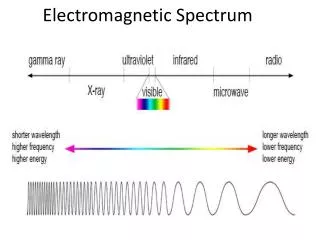
E = hv Where, E = energy of the photon (J) h = Planck’s constant (Js) v = frequency of the wave (Hz) E = hc/λ Where, E = energy of the photon (J) h = Planck’s constant (Js) c = speed of light, 2.99 x 108 m/s λ = wavelength of light (m) Important Equations and their units
When we see light from a neon sign, we are observing radiation from excited neon atoms. If this radiation has a wavelength of 640 nm, what is the energy of the photon being emitted? Planck’s Practice Problems
1. Light with a wavelength of 614.5 nm looks orange. What is the energy, in joules, of a photon of this orange light? 2. A photon of light produced by a surgical laser has an energy of 3.027 x 10 -19 J. Calculate the frequency and the wavelength of the photon. Planck’s Practice Problems
Wikipedia 2011, Max Planck, viewed 10 September 2011, < http://en.wikipedia.org/wiki/Max_Planck >. Wikipedia 2011, Planck’s Constant, viewed 10 September 2011, <http://en.wikipedia.org/wiki/Planck_constant>. Works Cited