Practice biochemistry amino acid
600 likes | 983 Views
Practice biochemistry amino acid. Introduction. Food are divided into three classes : 1- Carbohydrate Source of energy 2 - Lipid Principal of energy reserve 3 - Proteins Energy for growth and cellular maintance. Amino acid and protein.

Practice biochemistry amino acid
E N D
Presentation Transcript
Introduction • Food are divided into three classes : 1- Carbohydrate Source of energy 2- Lipid Principal of energy reserve 3- Proteins Energy for growth and cellular maintance
Amino acid and protein • Amino acid are the building block of proteins • There are about 300 amino acids occur in nature. Only 20 of them occur in proteins.
Structure of amino acids: • Proteins consists of amino acid linked to peptide bond • Each amino acid consists of : • Central carbon atoms • An amino acid • Carboxyl group • Side chain • Different side chain result in various amino acid
The carbon on all amino acids, except glycine, is a chiral carbonbecause it has four different groups bonded to it.
Glycine : Is simple amino acid because R chain is H
Proline • It is unique among the 20 protein-forming amino acids in that the amine nitrogen is bound to not one but two alkyl groups, thus making it a secondary amine
an imino acidis any molecule that contains both imino (>C=NH) and carboxyl (-C(=O)-OH) functional groups
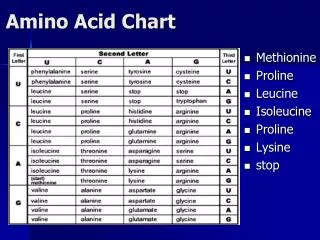
Essential Amino Acids in Humans • Required in diet • Humans incapable of forming requisite Arginine Histidine Isoleucine Leucine Valine Lysine Methionine Threonine Phenylalanine Tryptophan
Non-Essential Amino Acids in Humans • Not required in diet Alanine Asparagine Aspartate Glutamate Glutamine Glycine Proline Serine
Nonpolar amino acid • Hydrophobic amino acid: are amino acid that contain C,H in their side chain • They tend way from water (hate water) • Hydrophobic (normally buried inside the protein core):
Polar amino acid • also called hydrophilic (love water) • Tend to found on surface • That amino acid that contain in their side chain O,N and they can dissolve in water ( like dissolve like ) (covalent bond = hydrogen bond causing folding protein )
Uncommon Amino Acids • Hydroxylysine, hydroxyproline which is the building block of collagen • Carboxyglutamate which is the building block of blood-clotting proteins
At acidic pH, the carboxyl group is protonated and the amino acid is in the cationic form At neutral pH, the carboxyl group is deprotonated but the amino group is protonated. The net charge is zero; such ions are called Zwitterions At alkaline pH, the amino group is neutral –NH2 and the amino acid is in the anionic form.
Qualitative test for amino acids • There number of test to detect the presence of amino acid • This is largely depend on the natural of side chain
Ninhydrin Test • Ninhydrin is a chemical used to detect free amino acid and proteins • Amino acids(NH2) also react with ninhydrin at pH=4. • The reduction product obtained from ninhydrin then reacts with NH3 and excess ninhydrin to yield a blue colored substance. • This reaction provides an extremely sensitive test for amino acids.
With all amino acid will give purple or deep blue with exception Proline gives yellow not violet (why)
Proline reacts with ninhydrin, but in a different way. While most ninhydrin tests result in a purple color, the proline reaction is more yellow due to substitution of the alpha amino group that ninhydrin reacts with carbon rings
Procedure: • To 1 mL solution add 5 drops of 0.5% ninhydrine solution • Boil over a water bath for 2 min. • Allow to cool and observe the blue color formed.
biuret test : • Biuret structure: it is result of condensation of two molecule of urea
peptides containing three or more amino acid residues form a colored chelate complex with cupric ions (Cu2+) in an alkaline environment containing sodium potassium tartrate. • Single amino acids and dipeptides do not give the biuret reaction, but tripeptides and larger polypeptides or proteins will react to produce the light blue to violet complex that absorbs light at 540nm • One cupric ion forms a colored coordination complex with four to six nearby peptides bonds. The intensity of the color produced is proportional to the number of peptide bonds participating in the reaction..
Thus, the biuret reaction is the basis for a simple and rapid colorimetric reagent of the same name for quantitatively determining total protein concentration. • The working range for the biuret assay is 5-160mg/mL. • What is chelation? chelation involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central atom.
Principle: • The biuret reagent (copper sulfate in a strong base) reacts with peptide bonds in proteins to form a blue to violet complex known as the “biuret complex”. • N.B. Two peptide bonds at least are required for the formation of this complex.
Procedure: • To 2 ml of protein solution in a test tube, add 4ml of reagent incubation 30 min • Result :
Millons test : • It is specific for tyrosine, the only amino acid that contain a phenol group on which a hydroxyl group is attached. It gives red precipitate. • Consequently, any protein containing tyrosine will give a positive test of a pink to dark-red color.
Principle: • A test for protein, the tyrosine(phenolic group) of which reacts with nitrite after treatment with mercuric ion in acid to give a red color. • The reagent used in this test is called Millon's reagent and it contains mixture of Hg ++, Hg2 ++, HNO3 and HNO2
Procedure & observation: • To 2 ml of protein solution in a test tube, add 3 drops of Millon’s reagent. • Mix well and heat directly on a small flame. BWB 5 min • A white ppt is formed with albumin and casein (but not gelatin); • the ppt gradually turns into brick red.
Xanthoproteic Test: • Some amino acids contain aromatic groups that are derivatives of benzene. These aromatic groups can undergo reactions • One such reaction is the nitration of a benzene ring with nitric acid. The amino acids that have activated benzene ring can readily undergo nitration. • In the presence of activated benzene ring, forms yellowproduct. Apply this test to tyrosine, tryptophan, phenylalanine and glutamic acid.
Why benzene ring in phenylalanine is inactive • Phenylalanine still reacts with nitric acid, just not as readily as Tyr or Trp because Tyr and Trp have electron donors (hydroxyl and the nitrogen heteroatom), which make the ring a lot more attractive for electrophilic nitration. • To get Phe to react, you may have to boil it with nitric acid
Procedure: • To 2 mL amino acid solution in a boiling test tube, add equal volume of concentrated HNO3. • Heat over a flame for 2 min and observe the color. • Now COOL THOROUGHLY and CAUTIOSLY run in sufficient 3ml NaOH (why) • Observe the color of the nitro derivativative of aromatic nucleus.
Hopkins-Cole (Glyoxylic Acid Reaction) • Specific for tryptophan (the only amino acid containing indole group) • Reacting with a glyoxylic acid in the presence of a strong acid, the indole ring forms a violet cyclic product. • The protein solution is hydrolyzed by conc. H2SO4 at the solution interface. • Once the tryptophan is free, it reacts with glyoxylic acid to form violet product. Indole Glyoxylic acid
Procedure.. • In a test tube, add to 2 ml of the solution an equal volume of Hopkins- Cole reagent and mix thoroughly. • Incline the tube and let 5 to 6 ml of conc. H2S04 acid flow slowly down the side of the test tube, thus forming a reddish - violet ring at the interface of the two layers. That indicates the presence of tryptophan.
Sulfur test: Sulfur containing amino acids, such as cysteine and cystine upon boiling with sodium hydroxide (hot alkali) yield sodium sulfide. This reaction is due to partial conversion of the organic sulfur to inorganic sulfide, which can detected by precipitating it to lead sulfide, using lead acetate solution.S.(protein) + 2NaOH-------- Na2S Na2S + (CH3COO)2pb ------- PbS + 2CH3COONa
Procedure: 1. Place 1 ml of 2% casein, 2% egg albumin, 2% peptone, 2% gelatine and 0.1 M cysteine into separate, labeled test tubes. 2. Add 2 ml of 10 % aqueous sodium hydroxide. Add 5 drops of 10 % lead acetate solution. 3. Stopper the tubes and shake them. Remove the stoppers and heat in a boiling water bath for 5 minutes. Cool and record the results.
Sakaguchitest • For detection of the amino acid containing the guanidinium group (e.g. arginine). • In basic conditions, α- naphthol and sodium hypobromite/chlorite react with the guanidinium group to form redorange complexes.
Procedure: • 1. Add 1 ml of 3 N NaOH solution to 1 ml of the protein solution, followed by addition of 0.5 ml of 0.1 % α- naphthol solution, and a few drops of 2 % hypobromite solution (NaOBr). • 2. The formation of a red color indicates the presence of a guanidinium group in the compound under examination.