Galvanic corrosion
Galvanic corrosion.

Galvanic corrosion
E N D
Presentation Transcript
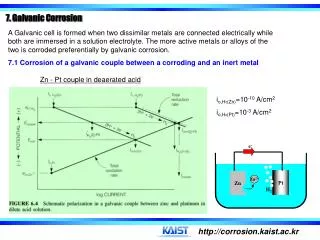
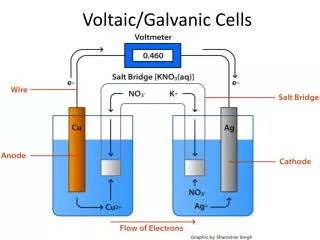
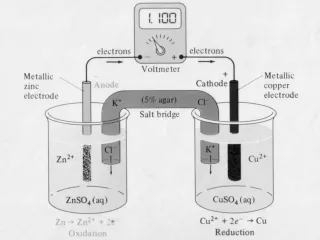
Galvanic corrosion • Galvanic corrosion is either a chemical or an electrochemical corrosion. The latter is due to a potential difference between two different metals connected through a circuit for current flow to occur from more active metal (more negative potential) to the more noble metal (more positive potential) • Galvanic coupling is a galvanic cell in which the anode is the less corrosion resistant metal than the cathode
Galvanic corrosion can be predicted by using the electromotive force (emf) or standard potential series for metal reduction listed in Table at the next slide • In selecting two metals or two alloys for a galvanic coupling, both metals should have similar potentials or be close to each other in the series in order to suppress galvanic corrosion
Galvanic coupling can be used for cathodic protection purposes
Other types of galvanic coupling are batteries and fuel cells • Lithium Ambient-Temperature Batteries (LAMBS) • Lead-Acid Battery • Dry-Cell Battery • Sintered Nickel Electrode in Alkaline Batteries
MICROSTRUCTURAL EFFECTS A mechanically deformed metal or alloy can experience galvanic corrosion due to differences in atomic plane distortion and a high dislocation density
Improper heat treatment can cause nonuniform microstructure and therefore, galvanic-phase corrosion is enhanced in corrosive media • Galvanic corrosion can occur in a polycrystalline alloys, such as pearlitic steels, due to differences in microstructural phases • In this case, pearlite consists of ferrite and cementite and when it is etched with a mild acid, which is the electrolyte, galvanic microcells between ferrite (cathode) and cementite (anode) are generated. Consequently, pearlite is revealed as dark cementite andwhite ferrite.
LOCALIZED CORROSION This term implies that specific parts of an exposed surface area corrodes in a suitable electrolyte. This form of corrosion is more difficult to control than general corrosion. • Crevice Corrosion which is associated with a stagnant electrolyte such as dirt, corrosion product, sand, etc. It occurs on a metal/alloy surface holes,underneath a gasket, lap joints under bolts, under rivet heads.
Filiform Corrosion is basically a special type of crevice corrosion, which occurs under a protective film. It is common on food and beverage cans being exposed to the atmosphere. • Pitting Corrosion is an extremely localized corrosion mechanism that causes destructive pits. • Oral Corrosion occurs on dental alloys exposed to saliva. • Biological Corrosion due to fouling organisms non-uniformly adhered on steel in marine environments. • Selective Leaching Corrosion is a metal removal process from the base alloy matrix, such as dezincification ( Zn is removed) in Cu-Zn alloys and graphitization (Fe is removed) in cast irons.
PITTING CORROSION • This form of corrosion is extremely localized and it manifests itself as holes on a metal surface. • The initial formation of pits is difficult to detect due to the small size, but it requires a prolong time for visual detection. • This form of corrosion can be found on aluminum and its alloys and automobile chromium-plated bumpers or body coated (painted) parts due to film/coating breakdown at isolated surface sites. • Pits vary in shape, but are very small surface holes due to the extremely localized anodic reaction sites
The initiation of pits occurs at localized sites on a metal surface defects, which may be due to coating failure, mechanical discontinuities or microstructural phase heterogeneities such as secondary phases. • The depth of pits can be found: d =λtn