Atom Notes
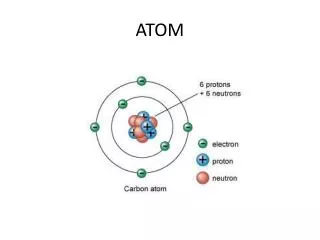
Atom Notes. Atom – small particle that makes up most types of matter (One million atoms lined up equals the thickness of one human hair) Atomic structure Nucleus – central part of an atom Protons – positively charged particles in the nucleus of an atom

Atom Notes
E N D
Presentation Transcript
Atom Notes Atom – small particle that makes up most types of matter (One million atoms lined up equals the thickness of one human hair) Atomic structure Nucleus – central part of an atom Protons – positively charged particles in the nucleus of an atom Neutrons – uncharged particles in the nucleus of an atom Electrons - negatively charged particles in an atom
History of Atomic Theory Dalton – (atomic model) Theory that substances were made of atoms, small, hard, dense spheres that could not be created, destroyed, or altered. Dalton’s model of the atom John Dalton
History of Atomic Theory Thomson – (discovering the electron) Proposed that atoms were made up of smaller particles. Theory that smaller negatively charged particles are spread evenly around a positively charged nucleus. His model was called the plum-pudding model.
History of Atomic Theory Rutherford – (the nucleus) Proposed that atoms had a dense, positively charged nucleus surrounded by electrons. Chadwick – discovered the neutrons Rutherford’s model
History of Atomic Theory Bohr – Said that electrons revolve around the nucleus in circular paths or orbits and that electrons could only exist in certain orbits and at certain energy levels. nitrogen Example of Bohr’s model of the atom
Periodic Table Mendeleev – arranged 63 elements into groups based on their chemical properties and atomic weights.
Periodic Table Hydrogen 1 H 1.008 Element name Atomic number Symbol State of matter Atomic mass
Element Information For any element: Number of Protons = Atomic Number Number of Electrons = Number of Protons = Atomic Number Number of Neutrons = Mass Number - Atomic Number Mass number = Protons + Neutrons (not found on periodic table)
Isotopes Isotopes have the same number of protons but a different mass number. Example: Carbon –12 and Carbon –14 Average atomic mass is an average of all the isotopes (can be found on the periodic table)
Metal, Nonmetal, Metalloid Metals – good conductors -malleable and ductile -lose electrons Nonmetals – poor conductors -brittle -gain electrons -form covalent bonds Metalloids – show properties of both metals and nonmetals.