Atom notes
Atom notes. Atom: basic unit of matter, the smallest part of an element that still has the element’s properties. Make a three flap foldable for subatomic parts. Sub atomic particles . http:// www.glencoe.com/sites/common_assets/science/virtual_labs/E18/E18.html atomic structure Protons

Atom notes
E N D
Presentation Transcript
Atom notes Atom: basic unit of matter, the smallest part of an element that still has the element’s properties.
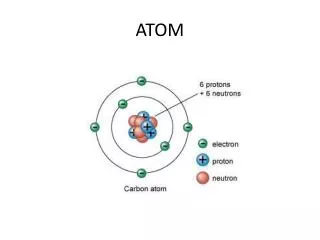
Sub atomic particles • http://www.glencoe.com/sites/common_assets/science/virtual_labs/E18/E18.html atomic structure • Protons • Positively charged • Electrons • Negatively charged • Neutrons • No charge http://web.buddyproject.org/web017/web017/images/atom.JPG http://www.eskom.co.za/nuclear_energy/fuel/atom.jpg
Protons • Positively charged particle • In the nucleus (center) of an atom
Neutron • Neutral (no) charge • In the nucleus of an atom
Electron • Negatively charged • Found on the outside of the nucleus in “orbitals” making up the “electron cloud”
http://ri-itest.portal.concord.org/preview/ • Physics activities • Atomic structure • Page 3 the elements
Protons: • The name of an atom is defined by its number of protons • The atomic number determines the name of an atom.
Example: Lithium 3 (+) protons and 3 (-) electrons Neutral – no charge 3 (+) protons and 2 (-) electrons +1 charge – positive ion Ion: An atom that has an unequal number of protons and electrons. Ions have a positive or negative charge Atoms that have a neutral charge are not ions Ion (has to do with electrons)
Ion explanation http://ri-itest.portal.concord.org/preview/ • Physics activities • Atomic structure • Page 4 ions
Isotope (has to do with neutrons) • Isotopes are different atoms with • the same number of protons • with different number of neutrons • This means the atomic mass of Helium isotopes are different.
Isotopes explanation http://ri-itest.portal.concord.org/preview/ • Physics activities • Atomic structure • Page 5 isotopes and radioactivity
Atomic number vs Atomic mass • Atomic Number • Number of protons • Atomic Mass/Mass number • Number of protons + neutrons • On the periodic table: it is an average mass
End of 3 flap foldable • Isotopes with people activity for average atomic mass • Do Atomic Model Food Lab • Take atomic structure and vocabulary quiz
Elements • Elements • a pure substance that consists entirely of one type of atom • Number of protons= element’s atomic# http://www.astro.virginia.edu/class/oconnell/astr121/im/periodic_table.gif
Periodic Table of Elements • Groups similar elements together • For each element gives: • Symbol • Atomic number • Average atomic mass • Group number, • period number, • metal or nonmetal
Chemical compounds • Compound – • Formed by the chemical combination of 2 or more elements in definite proportions • Example: C6H12O6 (glucose) http://www.biochem.arizona.edu/classes/bioc462/462bh2008/462bhonorsprojects/462bhonors2007/gsantarelli/experimentbackground.html
Covalent vs. Ionic chemical bonds • Ionic bonds • Electrons are transferred from one atom to another • Covalent bonds (strongest bond) • Electrons are shared between atoms • Forms molecules
Chemical formulas • H2O • 2 Hydrogen, 1 Oxygen • C6H12O6 • 6 carbon, 12 hydrogen, 6 oxygen • 6CO2 • 6 molecules of carbon dioxide, 1 carbon, 2 oxygen • = 6 carbon, 12 oxygen
Chemical Reactions • A process that changes one set of chemicals into another • Always involves the breaking and formation of bonds • Reactants • Enter into a reaction • Bonds are broken • Products • Result from a reaction • New bonds are formed Example Photosynthesis: 6H2O + 6 CO2C6H12O6 + 6O2
Changing Atomic Nuclei • Radioactivity • Fission and fusion
Radioactivity: a changing nucleus • Some atoms have unstable nuclei that undergo radioactive decay • Emit particles or release energy to become stable. • Radioactive atoms can • transform into a different isotope of the same element or • Transform into a different element. • The change is called radioactive decay
Molecular model dealing with radioactivityhttp://ri-itest.portal.concord.org/preview/ 1. QUESTION: When you used the model for making stable versus radioactive forms of elements, what was the general rule for creating an atom with a stable nucleus (one that is not radioactive)? ANSWER: The more equal the number of protons to the number of neutrons the more likely the atom will be stable. Also large nuclei are more likely unstable then small nuclei. 2 . True or False: Stable isotopes always have an equal number of protons and neutrons. ANSWER: FALSE, stable isotopes can have unequal protons and neutrons, for example Carbon 13 (6 protons, 7 neutrons) GO TO Physics activities Atomic structure Page 3 “Isotopes and Radioactivity”
Radioactivity: Good and Bad • BAD: Energy given off by radioactive matter can damage cells • When cells are exposed to nuclear radiation, the bonds holding together proteins and DNA molecules may break, and the cells may no longer function properly. • Good: Radioactive tracers • A radioactive isotope is taken up by an organism and becomes part of a chemical reaction which can then be traced by keeping track of where the radiation in the organism is coming from.
Half-life • Measurement of the amount of time it takes for ½ of a radioactive substance to decay. • Used to date how old substances are Rubidium Strontium dating is used for dating igneous rock and has a ½ life of 49 billion years. Carbon-14 used to date deceased organisms and has a ½ life of 5,700 years Radon: a gas that can collect in buildings and increase a persons chance of lung cancer has a ½ life 3.82 days Computer simulation of decay: http://www.lon-capa.org/~mmp/applist/decay/decay.htm
http://www.glencoe.com/sites/common_assets/science/virtual_labs/E18/E18.html half life simulator • http://phet.colorado.edu/en/simulation/radioactive-dating-game
Using radioisotopes for dating • Scientists can measure the amount of radioactive parent isotope and stable daughter isotope in a sample. Then scientists can use this ratio and the radioisotope’s half life to calculate the age of the sample.
assignments • Textbook chapter 10.1 Radioactivity, and 10.2 Rates of Radioactive Decay • “Radioactivity problems” • US geological survey article: “What is Radon”
Nuclear reactions: Fission vs. Fusion • Fission: nuclei of 1 atom is split to create 2 atoms this releases energy • Nuclear power plants • Nuclear bombs/weapons • Uranium and plutonium used http://www2.kutl.kyushu-u.ac.jp/seminar/MicroWorld3_E/3Part3_E/3P33_E/U235_fission_2_E.jpg
Fusion • Fusion: nuclei of 2 atoms are combined (fused) into 1 atom this releases energy • The sun is a natural fusion reactor. hydrogen hydrogen Helium http://knol.google.com/k/-/-/oml631csgjs7/e4w1oo/fusion.jpg
Video: Physical Science: Nuclear Energy united streaming. Com (20 min)
Video questions. • 1. The sun is a nuclear reactor because gravity in the nucleus makes hydrogen fuse to make helium and this releases tons of energy. • 2. Nuclear fission when the nucleus of an atom splits into two different atoms. Fission and fusion produce energy missing matter is transformed into energy. • 3. Some of the energy released is the type that damages cells.
4. Pierre and Maria Curie discovered radium and plutonium that give off energy. Irene Curie developed artificial radiation by making aluminum radioactive. • 5. The first atomic bomb was tested in 1945. It was used a couple months later by American to bomb Japan in WWII • 6. Hydrogen bombs are fusion, atom bomb are fission. The fusion releases more energy then fission. • 7. Nuclear submarines use Uranium to create electricity and don’t have to surface to refuel.
Enriching Uranium • Enriching uranium increases the amount of "middle-weight" and “light-weight” uranium atoms. Not all uranium atoms are the same. When uranium is mined, it consists of heavy-weight atoms (about 99.3% of the mass), middle-weight atoms (0.7%), and light-weight atoms (< 0.01%). These are the different isotopes of uranium, which means that while they all contain 92 protons in the atom’s center (which is what makes it uranium). The heavy-weight atoms contain 146 neutrons, the middle-weight contain 143 neutrons, and the light-weight have just 142 neutrons. To refer to these isotopes, scientists add the number of protons and neutrons and put the total after the name: uranium-234 or U-234, uranium-235 or U-235, and uranium-238 or U-238. • The fuel for nuclear reactors has to have a higher concentration of U-235 than exists in natural uranium ore. This is because U-235 is the key ingredient that starts a nuclear reaction and keeps it going. Normally, the amount of the U-235 isotope is enriched from 0.7% of the uranium mass to about 5%. Gaseous diffusion is the only process being used in the United States to commercially enrich uranium. Gas centrifuges can also be used to enrich uranium. http://www.nrc.gov/materials/fuel-cycle-fac/ur-enrichment.html
Uranium enrichment: Separate the isotopes to get the U-235 http://www.nrc.gov/materials/fuel-cycle-fac/ur-enrichment.html
Extra info News article Kepler 22B discovered a habitable planet Dec 5, 2011 NPR • http://www.npr.org/2011/12/05/143142279/found-earth-like-planet-that-might-be-right-for-lifeFound: Earth-Like Planet that might be right for life • Heard on All Things Considered National Public Radio • December 5, 2011 - ROBERT SIEGEL, HOST: • What is the name of the space telescope found the interesting planet? • What evidence did scientists see to prove the planet exists? • What does the “habitable zone” mean? • One scientist says we should “discard the habitable zone” and look for other planetary characteristic instead?
Extra- in depth material: Electron • Orbital’s: the regions in an atom where electrons are likely to be found. • Electrons with different amounts of energy are found different distances from the nucleus. • Electrons occupy the lowest energy level orbital’s first. http://course1.winona.edu/sberg/ILLUST/per-chrt.gif
Extra- in depth material: Electron: Orbitals have complex shapes • Because electrons repel each other the shapes of the orbital’s are complex shapes. • Four names of orbital’s are s, p, d, and f.