Topic 7 Heat Transfer
Topic 7 Heat Transfer. FOOD1360/1577 Principles of Food Engineering Robert Driscoll. Heat Transfer. S&H Chapter 4 Notes on electric circuits Sect 3.2. Movement of heat energy from one point to another by virtue of temperature gradient (difference). Heat flux. hot. cold.

Topic 7 Heat Transfer
E N D
Presentation Transcript
Topic 7Heat Transfer FOOD1360/1577 Principles of Food Engineering Robert Driscoll FST 2010
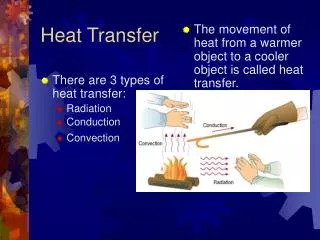
Heat Transfer S&H Chapter 4 Notes on electric circuits Sect 3.2 • Movement of heat energy from one point to another by virtue of temperature gradient (difference) Heat flux hot cold FST 2010
Use in unit operations: • Used in most unit operations in food industry • Why do we heat? • Cooking • Sterilising • Pasteurising • Blanching • Browning • Why do we cool? • Preservation • Texture (icecream!) • Stop cooking quickly (see S&H Sect 4.1: Heat Exchangers). FST 2010
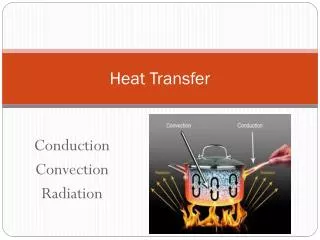
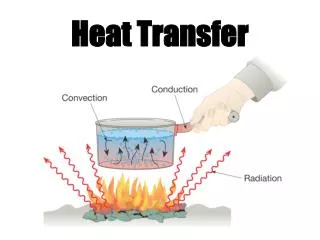
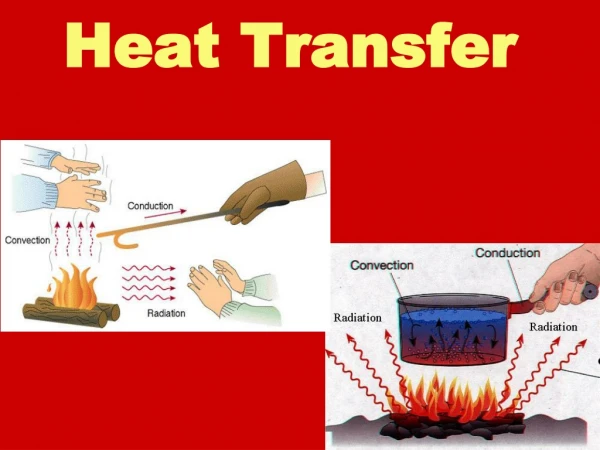
Modes of heat transfer • Conduction • Convection • Radiation These notes follow S&H Sect 4.3. So look at his examples! FST 2010
1. Conduction • Heat transfer between neighbouring molecules Esp. for solids, but also for fluids!·vibration of molecules, or·drift of electrons (in metals)·no net movement of molecules FST 2010
Fourier’s equation • Rate of flow = driving force / resistance • The negative indicates heat flows from hot to cold. • Logical: rate of heat flow depends on • Temperature difference • Material properties • Distance to flow • Cross-sectional area for flow. FST 2010
Terms used • dQ/dt = rate of heat flow (J/s or Watts) • dx = thickness (m). Use Dx for finite differences. • A = area normal to the flow direction (m2) • k = thermal conductivity of the material (in J/oC.s.m or W/m.K) • dT = temperature difference (oC) (use DT = T1-T2 for finite difference). • For steady state heat flow through a material, FST 2010
Example: slab A T1 k T2 x FST 2010
Worked Example • Use Fourier’s equation to findDT=T1-T2 (see pic). • Solution: • Why is Dx negative? A=0.1m2 T1 T2 k=0.2 x=25 mm FST 2010
Thermal Conductivity, k • thermal conductivity: a property of a material • good conductors of heat: metals • bad conductors of heat: insulators • What are typical values? FST 2010
Some examples Metals: 50-400 J/oC.s.m Glass: 0.51 J/oC.s.m Alloys: 10-120 J/oC.s.m Air: 0.024 J/oC.s.m Water: 0.7 J/oC.s.m Ice: 2.3 J/oC.s.m Insulations: 0.026-0.052 J/oC.s.m (cork, foamed plastics, expanded rubber etc.) • Air is a good insulator! FST 2010
Thermal conductivity of Foods • Close to k for water for higher moisture foods • For fruits and vegs with W >60%: k = 0.148 + 0.00493 W W = water content in % • Meats with W=60-80%: k = 0.08 + 0.0052 W • For solid foods, low W, see Toledo, Singh and Heldman, Fellows etc. FST 2010
Example • What is the thermal conductivity of a block of meat with a moisture content of 75%? • How would this information be useful? FST 2010
Notes: • Heat flux = heat transfer rate per unit area = dQ/Adt (in J/s.m2) • Use dx for differential, x for difference. • Power is measured in Watt (W) • Energy is measured in Joule (J) • Power is energy per second (W = J/s) FST 2010
Example • A cork slab 10 cm thick has one face at -12oC and the other at 21oC. If the thermal conductivity of cork is 0.042 W/mK, estimate the heat transfer per square meter. Solution: ‘Per square meter’ means find heat flux. So which gives 13.9 W/m2. FST 2010
Similarity to electrical circuits • From physics - electrical circuits: • heat flow corresponds to electric current • x/kA corresponds to resistance • T corresponds to voltage (driving force) • This is a useful analogy. FST 2010
Electrical Circuit • For a simple circuit, V A R I FST 2010
dT R=dx/kA Apply to heat So we have defined a heat resistance, R FST 2010
dT k1 k2 T1 T2 dx1 dx2 Flow through layers • If heat flows through a series of layers of material, we call it series heat transfer. REAL ANALOGY FST 2010
With the electric circuit, total resistance is R1 + R2. • So total heat resistance is: • Setting RT=1/UA is convenient: • U is the overall heat transfer coefficient. Defines heat flowresistance. Defines heat flowin terms of U FST 2010
2. Convection • Heat transfer by the net movement of moleculesin a fluid is called a convection current: Fluid Flow Cold Body Hot Body FST 2010
Hot Body Ts Fluid Flow T Just for one surface • Governing equation: FST 2010
Two Types of Convection • Natural convection (slow heat transfer): • induced by temperature gradient • current driven by density difference • Forced convection (rapid heat transfer): • movement of molecules by external energy • e.g. a stirrer, fan, agitator, pump etc. FST 2010
Which is faster? • In liquids, convection is much faster than conduction! Soup heats faster than dog food. ConvectionMixing HeatDiffuses FST 2010
Newton’s law of cooling • where • h = convective heat transfer coefficient • A = area of heat transfer • T= temperature difference. • h is also called the surface heat transfer coefficient (J/oC.s.m2 or W/m2K) FST 2010
Convective resistance • Since dQ/dt=hAT, then • Again comparing with our electrical circuit analogy: • we can identify a convective resistance as R=1/hA. FST 2010
Examples of htc • ‘htc’ = heat transfer coefficient • Values determined experimentally • Air: Free convection 5-25 J/oC.s.m2 Forced convection 10-200 • Water: Free convection 20-100 Forced convection 50-10,000 Boiling water 3,000 –100,000 Condensing water vapour 5000 – 100,000 FST 2010
Example • Estimate the rate of heat loss from a hot plate with a surface area of 1m2. The plate surface temperature is 120oC and the ambient temperature is 20oC. Convective heat transfer is estimated to be 10J/oC.s.m2 FST 2010
3. Radiation • heat transfer by electromagnetic waves • similar to transfer of light • Important in baking ovens • requires no physical medium for propagation • can occur in vacuum (unlike conduction and convection) FST 2010
Radiation equation q = s e A T4 • where • s = Stefan-Boltzmann constant • = 5.669x10-8 J/m2.s.K4 • e = emissivity, relative to a black body • T = temperature of emitting body in K FST 2010
Example calculation • Calculate the rate of heat energy emitted by 10 m2 of a polished stainless steel tube (emissivity=0.15). The temperature of the tube is 100oC. FST 2010
R1 R2 R3 Combined modes • For resistances in series, add resistances. h2,A h1,A k,x,A T1 T2 FST 2010
Finding U: • Adding the resistances: • Solving: • where U is the overall htc for the system. Note: UA is the reciprocal of RT FST 2010
100oC base 3 mm 250oC Worked Problem • Water boiling in a Cu saucepan (ID=200 mm). • For copper, k=400 W/m.K • Convective resistance (base to water) = 0.0105 K/W • Find rate of heat transfer. FST 2010
Solution • Two resistances: • conduction through copper saucepan base • convection from base to water. • So find each resistance, then add: • So heat flow is: FST 2010 Later problem: other types
Heat Flow Thru Pipes Exterior, T2 Length, L Product, T1 r2 r1 FST 2010
End View • Apply Fourier’s equation to heat flows through a thin annulus, dr. dr T2 r2 r1 T1 FST 2010
Derivation • Since FST 2010
Solving: FST 2010
Cylinder thermal resistance • Comparing with the electrical circuit analogy again, we can identify Rcyl: • This resistance acts like the slab and convective resistances seen before. FST 2010
150oC Example: Two Conductive Layers • Calculate the rate of heat loss through a fibreglass insulated steam pipe. Ignore convection. fibreglass T=35oC ID = 2.1 cm MD = 2.67 cm OD = 3.335 cm kfibreglass= 0.0351 W/m.K kpipe= 45 W/m.K iron pipe FST 2010
Solving for heat flow Two layers, two resistances to add. Rpipe= ln(1.335/1.05)/(2 45) (per unit length) = 0.00085 Rinsul= ln(1.667/1.335)/(2 0.0351) = 1.007 Thus rate of heat flow = (150-35)/(1.008) = 114 W/m Total resistance FST 2010
Example: Conduction / Convection • Data: h1=6.5 W/m2K h2=4.5 W/m2K T1=135oC T=20oC k1=202 W/mK ri=4.88 cm ro=5.08 cm h2, T k h1, T1 ri ro FST 2010
Solution • Inner convective resistance: • Outer convective resistance: • Pipe wall conductive resistance: FST 2010
So total resistance = 0.502+0.696+0=1.20 • So heat loss is: • Note that heat losses are written per unit length. Why? FST 2010
Insulation • Insulation has low thermal conductivity • Often used for lagging – covering pipes and vessels to reduce heat flow • Important over • long lengths or • large surface areas. FST 2010
Choice of insulation • Depends on: • Cost of materials vs. rate of heat loss • Conductivity of the insulation (may vary with age) • The finish (coating) of the insulation • Critical thickness! FST 2010
Critical Thickness • Adding insulation may increase heat losses! • Why? – increase in surface area. • where r0 is outer radius – main variable. FST 2010
Minimum RT • Differentiate wrt ro: • Solving, RT has a minimum at outer radius rc=k/h FST 2010
Plotting RT K/W FST 2010