HEATING AND COOLING CURVES
HEATING AND COOLING CURVES . Concepts. What are some things that happen as we heat a sample up?. Solid Liquid Gas Melting, Evaporating Increase in entropy. What is heat? How is heat different from temperature? . Heat is a measure of kinetic energy

HEATING AND COOLING CURVES
E N D
Presentation Transcript
HEATING AND COOLING CURVES Concepts
What are some things that happen as we heat a sample up? • Solid Liquid Gas • Melting, Evaporating • Increase in entropy
What is heat? How is heat different from temperature? • Heat is a measure of kinetic energy • In chemistry heat is measured in Joules (J) • Heat is dependent on mass, temperature change and specific heat • Temperature is a measure of average kinetic energy • Temperature is measured in Kelvin (K) or Celsius (C)
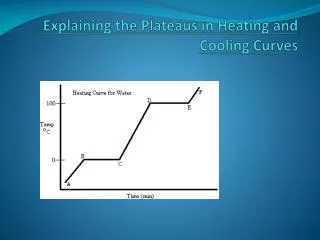
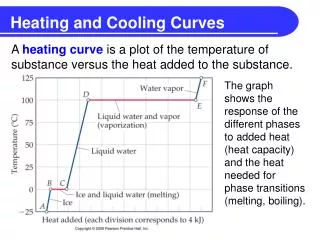
What is the heating curve? • The heating curve is a graph which represents how a sample changes phases. As heat is added over time, the sample changes temperature and phase accordingly. Thus heating curve.
Why is the curve flat at some portions? • Temperature is staying constant, but potential energy is increasing • During this time the solid completely becomes liquid
Why is the curve flat at some points? • This is called the heat of fusion (Hf) in segment BC and heat of vaporization (Hv) in segment DE. • Remember fusion is melting and vaporization is evaporation
What is heat of fusion? What is heat of vaporization? • Hf is the amount of energy needed to completely make a solid into a liquid • Hv is the amount of energy needed to completely make a liquid into a gas
Summary • The heating curve is a useful tool to show us the changes in temperature, energy, and entropy as a sample is heated up. It give us detailed information about phases and phase changes of samples.