Chapter 21 Amines
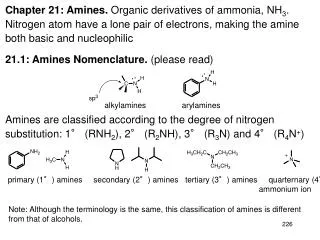
Chapter 21 Amines. Amine Nomenclature. Classification of Amines. Alkylamine N attached to alkyl group Arylamine N attached to aryl group Primary, secondary, or tertiary determined by number of carbon atoms directly attached to nitrogen. Nomenclature of Primary Alkylamines (RNH 2 ).

Chapter 21 Amines
E N D
Presentation Transcript
Classification of Amines • Alkylamine • N attached to alkyl group • Arylamine • N attached to aryl group • Primary, secondary, or tertiary • determined by number of carbon atoms directly attached to nitrogen
Nomenclature of Primary Alkylamines (RNH2) • Two IUPAC styles • 1) analogous to alcohols: replace -e ending by -amine • 2) name alkyl group and attach -amine as a suffix
NH2 CH3CHCH2CH2CH3 NH2 Examples: some primary alkylamines (RNH2: one carbon directly attached to N) ethylamine or ethanamine CH3CH2NH2 cyclohexylamine orcyclohexanamine 1-methylbutylamine or2-pentanamine
NH2 Br CH2CH3 F NH2 Nomenclature of Primary Arylamines (ArNH2) • Name as derivatives of aniline. p-fluoroaniline 5-bromo-2-ethylaniline
O HC NH2 Amino groups as substituents • amino groups rank below OH groups and higher oxidation states of carbon • in such cases name the amino group as a substituent HOCH2CH2NH2 2-aminoethanol p-aminobenzaldehyde
Secondary and Tertiary Amines • Name as N-substituted derivatives of parent primary amine. • (N is a locant-it is not alphabetized, but is treated the same way as a numerical locant) • Parent amine is one with longest carbonchain.
NHCH2CH3 NO2 Cl CH3 N CH3 Examples CH3NHCH2CH3 N-methylethylamine 4-chloro-N-ethyl-3-nitroaniline N,N-dimethylcycloheptylamine
CH3 – + – Cl N CF3CO2 CH2CH3 H Ammonium Salts • A nitrogen with four substituents is positivelycharged and is named as a derivative of ammonium ion (NH4+). + CH3NH3 methylammoniumchloride N-ethyl-N-methylcyclopentylammoniumtrifluoroacetate
CH3 + – CH3 CH2 I N CH3 Ammonium Salts • When all four atoms attached to N are carbon,the ion is called a quaternary ammonium ion andsalts that contain it are called quaternaryammonium salts. benzyltrimethylammonium iodide
Alkylamines 147 pm 112° 106°
Alkylamines Most prominent feature is high electrostaticpotential at nitrogen. Reactivity of nitrogen lonepair dominates properties of amines.
Geometry at N Compare geometry at N of methylamine, aniline,and formamide. • Pyramidal geometry at sp3-hybridized N in methylamine. • Planar geometry at sp2-hybridized N in formamide. H H H sp3 sp2 NH2 C NH2 C O H
Geometry at N Compare geometry at N of methylamine, aniline,and formamide. • Pyramidal geometry at sp3-hybridized N in methylamine. • Planar geometry at sp2-hybridized N in formamide. sp3 sp2
Geometry at N Angle that the C—N bond makes with bisector ofH—N—H angle is a measure of geometry at N. • Note: this is not the same as the H—N—H bond angle sp3 sp2 180° ~125°
Geometry at N Angle that the C—N bond makes with bisector ofH—N—H angle is a measure of geometry at N. sp3 sp2 180° ~125° 142.5°
Geometry at N Geometry at N in aniline is pyramidal; closer tomethylamine than to formamide. 142.5°
Geometry at N • Hybridization of N in aniline lies between sp3 and sp2. • Lone pair of N can be delocalized into ring best if N is sp2 and lone pair is in a p orbital. • Lone pair bound most strongly by N if pair is in an sp3 orbital of N, rather than p. • Actual hybridization is a compromise that maximizesbinding of lone pair. 142.5°
Electrostatic Potential Maps of Aniline Nonplanar geometry at N. Region of highestnegative potential is at N. Planar geometry at N. High negative potential shared by N and ring.
Physical Properties • Amines are more polar and have higher boiling points than alkanes; but are less polar andhave lower boiling points than alcohols. CH3CH2CH3 CH3CH2NH2 CH3CH2OH dipolemoment (): 0 D 1.2 D 1.7 D boiling point: -42°C 17°C 78°C
Physical Properties CH3CH2CH2NH2 CH3CH2NHCH3 (CH3)3N boilingpoint: 50°C 34°C 3°C • Boiling points of isomeric amines decrease ingoing from primary to secondary to tertiary amines. • Primary amines have two hydrogens on N capable of being involved in intermolecular hydrogen bonding. Secondary amines have one. Tertiary amines cannot be involved in intermolecular hydrogen bonds.
Effect of Structure on Basicity • 1. Alkylamines are slightly stronger bases than ammonia.
Table 22.1 (page 920)Basicity of Amines in Aqueous Solution • Amine Conj. Acid pKa • NH3 NH4+ 9.3 • CH3CH2NH2 CH3CH2NH3+ 10.8 CH3CH2NH3+ is a weaker acid than NH4+;therefore, CH3CH2NH2 is a stronger base than NH3.
Effect of Structure on Basicity • 1. Alkylamines are slightly stronger bases than ammonia. • 2. Alkylamines differ very little in basicity.
Table 22.1 (page 920)Basicity of Amines in Aqueous Solution • Amine Conj. Acid pKa • NH3 NH4+ 9.3 • CH3CH2NH2 CH3CH2NH3+ 10.8 • (CH3CH2)2NH (CH3CH2)2NH2+ 11.1 • (CH3CH2)3N (CH3CH2)3NH+ 10.8 Notice that the difference separating a primary,secondary, and tertiary amine is only 0.3 pK units.
Effect of Structure on Basicity • 1. Alkylamines are slightly stronger bases than ammonia. • 2. Alkylamines differ very little in basicity. • 3. Arylamines are much weaker bases than ammonia.
Table 22.1 (page 920)Basicity of Amines in Aqueous Solution • Amine Conj. Acid pKa • NH3 NH4+ 9.3 • CH3CH2NH2 CH3CH2NH3+ 10.8 • (CH3CH2)2NH (CH3CH2)2NH2+ 11.1 • (CH3CH2)3N (CH3CH2)3NH+ 10.8 • C6H5NH2 C6H5NH3+ 4.6
H •• + N H + H2N Strongeracid Strongerbase pKa = 4.6 H + H3N •• + NH2 Weakerbase Weakeracid pKa =10.6 Decreased basicity of arylamines
H N H H + H3N •• + NH2 Decreased basicity of arylamines •• + + H2N Strongeracid When anilinium ion loses a proton, theresulting lone pair is delocalized into the ring. Weakeracid
H N H Strongerbase H + H3N •• + NH2 Weakerbase Decreased basicity of arylamines •• + + H2N Aniline is a weaker base because its lone pair is more strongly held.
pKa of conjugate acid: 4.6 0.8 ~-5 Decreased basicity of arylamines • Increasing delocalization makes diphenylamine a weaker base than aniline, and triphenylamine a weaker base than diphenylamine. C6H5NH2 (C6H5)2NH (C6H5)3N
X NH2 Effect of Substituents on Basicity of Arylamines • 1. Alkyl groups on the ring increase basicity, but only slightly (less than 1 pK unit). X pKa of conjugate acid H 4.6 CH3 5.3
X NH2 Effect of Substituents on Basicity of Arylamines • 2. Electron withdrawing groups, especially ortho and/or para to amine group, decrease basicity and can have a large effect. X pKa of conjugate acid H 4.6 CF3 3.5O2N 1.0
– •• •• O O •• •• •• + + + •• N NH2 N NH2 O O •• •• •• •• – – •• •• p-Nitroaniline • Lone pair on amine nitrogen is conjugated with p-nitro group—more delocalized than in aniline itself. Delocalization lost on protonation.
Effect is Cumulative • Aniline is 3800 times more basic thanp-nitroaniline. • Aniline is ~1,000,000,000 times more basic than 2,4-dinitroaniline.
•• N N •• H piperidine pyridine pKa of conjugate acid: 11.2 pKa of conjugate acid: 5.2 (resembles anarylamine inbasicity) (an alkylamine) Heterocyclic Amines is more basic than
N H •• N •• N •• Heterocyclic Amines is more basic than imidazole pyridine pKa of conjugate acid: 7.0 pKa of conjugate acid: 5.2
N H •• N •• + H H N H N •• N N •• H Imidazole • Which nitrogen is protonated in imidazole? H+ H+ +
N H •• N •• + H H N H N H N N •• Imidazole • Protonation in the direction shown gives a stabilized ion. H+ + ••
Phase-Transfer Catalysis • Phase-transfer agents promote the solubility ofionic substances in nonpolar solvents. Theytransfer the ionic substance from an aqueousphase to a non-aqueous one. • Phase-transfer agents increase the rates ofreactions involving anions. The anion is relativelyunsolvated and very reactive in nonpolar mediacompared to water or alcohols.
CH2CH2CH2CH2CH2CH2CH2CH3 CH2CH2CH2CH2CH2CH2CH2CH3 H3C N CH2CH2CH2CH2CH2CH2CH2CH3 Phase-Transfer Catalysis Quaternary ammonium salts are phase-transfercatalysts. They are soluble in nonpolar solvents. + Cl– Methyltrioctylammonium chloride
CH2CH3 + CH2CH3 N CH2CH3 Phase-Transfer Catalysis Quaternary ammonium salts are phase-transfercatalysts. They are soluble in nonpolar solvents. Cl– Benzyltriethylammonium chloride
Example The SN2 reaction of sodium cyanide with butylbromide occurs much faster when benzyl-triethylammonium chloride is present than whenit is not. + CH3CH2CH2CH2Br NaCN benzyltriethylammonium chloride + CH3CH2CH2CH2CN NaBr
CH2CH3 + CH2CH3 N CH2CH3 CH2CH3 + + Cl– CN– CH2CH3 N (aqueous) CH2CH3 (aqueous) Mechanism + CN– Cl– (aqueous) (aqueous)
CH2CH3 + CH2CH3 CN– N CH2CH3 (in butyl bromide) CH2CH3 + CH2CH3 N CH2CH3 Mechanism CN– (aqueous)