Atomic Theory
Discover the evolution of atomic theory in this chapter. Beginning with Democritus's concept of indivisible units in the 4th century BCE, we navigate through Dalton's proposition that all atoms of an element are identical, followed by Thomson's discovery of electrons and the plum-pudding model. We also delve into Rutherford's identification of the atomic nucleus and Bohr's energy levels for electrons. Learn about the complexities of modern quantum theory, electron behavior, and the significance of valence electrons in determining chemical properties.

Atomic Theory
E N D
Presentation Transcript
Atomic Theory Chapter 4 Section 1
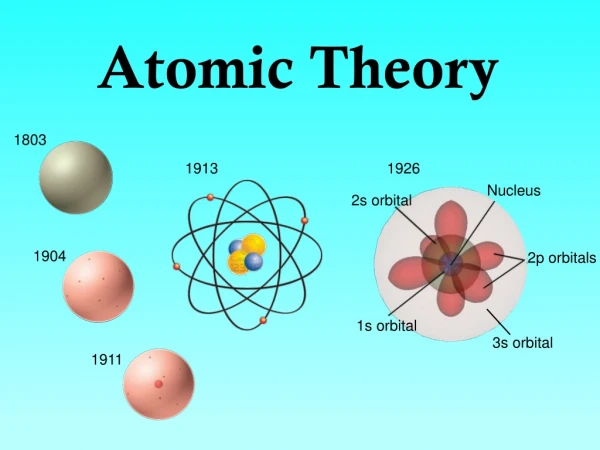
Now, we know that all matter is made up of particles called atoms • However, in the fourth century BCE, Democritus, the Greek philosopher, suggested that the universe was made of individual units Beginning of the Theory
Atom comes from the Greek term, atomos, which means “unable to be cut or divided” Greek meaning
In 1808, the first scientific atomic theory was proposed. Dalton, a schoolteacher, proposed that all atoms of a given element were exactly alike, and atoms of a element could join to form compounds. • Oxygen and Hydrogen in Water Dalton’s Atomic Theory
J.J. Thomson, a British scientist conducted an experiment to suggest that atoms were not inseparable in 1897. • While experimenting with cathode rays, Thomson suggested that the ray was constructed from a negatively charged particles from inside the atom. Thomson’s Model of the Atom
Thomson was responsibly for the discovery of an electron. • An electron is a negatively charged particle from the inside of an atom. • His model was called the plum-pudding model, because he stated that the electrons were spread throughout the atom like blueberries in a muffin. Discovery!
Rutherford, another British scientist, contradicted Thomson’s theory. • He stated that the mass of the atom was concentrated at the center of the atom. Rutherford’s Model
From firing positively charged particles at a gold foil, he was able to determine that all atoms contain a nucleus. • Nucleus is a positively charged, dense core of an atom. Rutherford’s Discovery
Modern Cell Theory Chapter 4 Section 3
Electrons can be found only in certain energy levels, not between levels. Furthermore, the location of electrons cannot be predicted precisely. Modern Theory
Neils Bohr, a Danish physicist, suggested that energy of each electron was related to the electron’s path around the nucleus in 1913. • Electrons can only be in certain energy levels and electrons gain or lose energy to move energy levels. Electrons location is limited
Bohr’s model of the atom no longer explained all the aspects of electron behavior. • It was not assumed that electrons orbited a nucleus like a planet, but actually acted like a wave. Electrons act like waves
Just like Where’s Waldo, electrons cannot be found. • Imagine a blade on a plane, you know that those blades are in the blurry part, but can you pick out each one? • This is like a electron, you know that they are present in the blurry part, but you cannot pick out where. Where is the electron?
The shaded part of the blade of a plane for an atom would be called the Orbit. • Orbit is the region where the electron can likely be found. Orbit
The number of energy levels that are filled in an atom depends on the number of electrons. • Valence Electrons are the electrons in the outer energy level of an atom. Valence Electrons determine the chemical properties of an atom. • Look at Periodic Table Electron Energy Levels
32 Electrons 18 Electrons 8 Electrons 2 Electrons
Four types: s, p, d, and f • S is the simplest kind of orbit, the shape of a sphere. This energy level can only hold two electrons. • P is the shape of a dumbbell and can orient in space in three different ways. One the y, x, and z axis. • Each p orbit can hold 2 electrons, so all together the three orbits can hold ____ electrons. Types of Orbits
The d and f orbits are more complex. There are five possible d orbits and seven possible f orbits. The shapes of these are completely different, but just like the other orbits can only hold 2 electrons. Types of Orbits
Orbits determine the number of electrons that each level can hold.
Electrons can jump energy levels, like we ride an elevator. • When an electron is at the lowest energy state it is called the ground state. • When an electron gets excited it gains a particle of light called a photon. Electron Transitions