Atomic Theory
Atomic Theory. History of the Discoveries Related to the Atom Mr. Addison. Timeline of Atomic Theory. 450 BC 1800’s _________________________________________________________. Democritus: matter was made of “atoms”.

Atomic Theory
E N D
Presentation Transcript
Atomic Theory History of the Discoveries Related to the Atom Mr. Addison
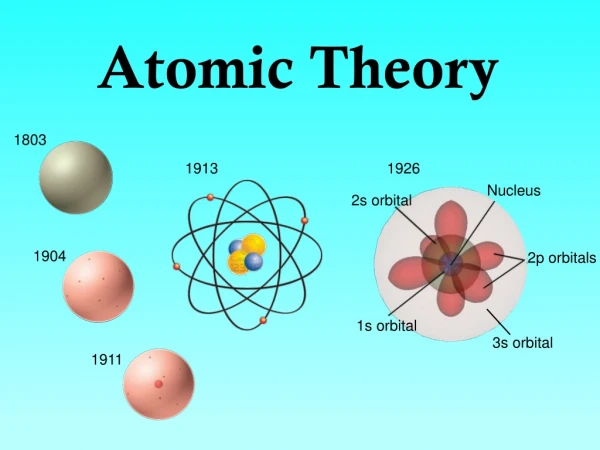
Timeline of Atomic Theory 450 BC 1800’s _________________________________________________________ Democritus: matter was made of “atoms” Dalton: proposed 5 components to an Atomic Theory.
Dalton’s Theory: Foundation of Future Investigations • All matter consists of tiny particles. • Atoms are indestructible and unchangeable. • Elements are characterized by the mass of their atoms. • When elements react, their atoms combine in simple, whole-number ratios. • When elements react, their atoms sometimes combine in more than one simple, whole-number ratio.
Atomic Theory 1897-1920 ___________________________________ Planck quanta 1901 Bohr quantum model 1913 Rutherford nucleus 1911 Thompson negative charge in the atom 1897
Contributions to Modern Atomic Theory De Broglie Wave nature of matter 1924 _____________________________________ Gell-Mann Quarks 1962 Heisenberg e- is a wave and a particle 1926 Schrodinger e- orbits as a wave 1926 Chadwick neutron 1932
Experiments that Characterized Particles of the Atom • Thompson Cathode Ray tube: • Electrons are negative in charge • http://chemmovies.unl.edu/ChemAnime/CATHRAYD/CATHRAYD.html • Rutherford’s Gold Foil: • Nucleus contains protons and is very small/dense • http://www2.wwnorton.com/college/chemistry/gilbert/tutorials/ch3.htm • Bohr model: • Electrons travel in certain paths • http://chemmovies.unl.edu/ChemAnime/BOHRQD/BOHRQD.html
Particles of the Atom to date: • Nucleus • Protons = p+ = 3 quarks • Neutrons= n0 = 3 quarks • Electron Cloud • Electrons =e- (particle/wave) Held together by a force called : Gluon
Types of Quarks Determine Chargehttp://nobelprize.org/educational_games/physics/matter/1.html Quarks = up (+2/3) and down (-1/3) p+ = 2ups and 1 downn0 = 2 down and 1 up +2 +2-1 = +1 -1-1+2 = 0 3 3 3 3 3 3 Gluon forces between quarks
Predicting the Location of an Electron in an Atom: • Bohr proposed 4 characteristics to consider to predict the location of an electron. • l = the distance from the nucleus • m = the shape of the orbital in which the e- occupies. (s,p,d,f) • n = axis the orbital occupies. (x, y, z) • s =spin of the e- (+1/2 , -1/2)
Distance from the nucleus: • Levels 1-7: When looking at the periodic table, can be determined by the period number. 1 2 3 4 5 6 7
Shape of the Orbital on the x,y,z Axis • The number of electrons in a particular region determines the shape. s shape 2e- p shape 6e-
Shape of the Orbital on the x,y,z Axis d shape 10e- f shape 14e-
Spin of the Electron • http://www.colorado.edu/physics/2000/applets/a2.html
Do Electrons behave as a particle or as wave? Definition of Light: • http://chemmovies.unl.edu/ChemAnime/DEFLITD/DEFLITD.html
Einstein and Bohr Bohr: -Predictability Einstein: -causality
Electron behavior as it moves to other energy levels. • This emitted energy is equal to the difference between the high and low energy levels, and may be seen as light. Light emission tutorial http://www2.wwnorton.com/college/chemistry/gilbert/tutorials/ch3.htm
Electrons are Particles / Photons • Electrons travel in discreet packets called photons. • To describe these unique packets or quantities of energy the term “quanta” is used. • Every element on the periodic table is made up of atoms with a unique signature of spectral lines
Electrons exhibit particle and wave like behavior“Duality”