Neutralizing an Acid
This experiment aims to determine the concentration of acetic acid in vinegar through titration with sodium hydroxide. Titration is a valuable technique for quality control in various fields, including forensic science and pharmaceuticals. By measuring a 25ml sample of vinegar, adding 2-4 drops of phenolphthalein indicator, and using a burette for sodium hydroxide, we can observe the neutralization reaction when the solution changes color. The collected data will allow for calculations to verify if the vinegar's acid concentration meets expected standards.

Neutralizing an Acid
E N D
Presentation Transcript
Neutralizing an Acid Aim To be able to calculate the concentration acid in vinegar.
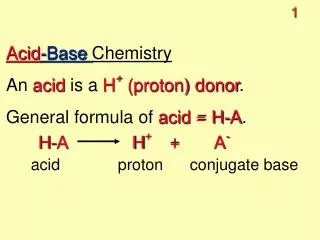
Context Titration – this is a technique that can be used to find out the concentration of a solution. It can be used for quality control, by forensic scientists and pharmacists to make medicines. You will be given a sample of vinegar and need to check if it has been watered down or not. You will be told the concentration of ethanoic acid it is supposed to contain and must check to see if it is correct.
Risk Assessment
Equipment Diagram
Method • Using a pipette, measure out 25ml of vinegar. • 2. Add 2-4 drops of phenolphthalein • indicator to the acid. • 3. Fill the burette to the 0ml mark with sodium • hydroxide. • 3. Turn the stop-cock on the burette and • release the sodium hydroxide until the vinegar • turns pink. • Record the sodium hydroxide reading in a table.
Analysis Neutralization happens when… The word equation for this reaction is… The balanced symbol for this reaction is… The concentration of ethanoic acid in vinegar was… The calculation to show this is…
Analysis and Evaluation To calculate the concentration of ethanoic acid in the vinegar you need to: Calculate the moles of sodium hydroxide used= volume in dm3 x concentration Use this value to work out concentration of ethanoic acid = moles volume in dm3
Evaluation My experiment went… I did not have problems with… because… I had problems with … because… To improve my experiment I could… My results would be more reliable if I… Did you have any results that look out of place? (Was the amount of sodium hydroxide needed to neutralize the acid very high or very low?) If so, how might this have happened?