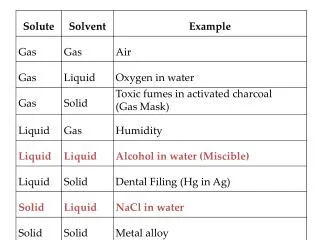
Solubility of Various Solutes in Water vs. Temperature
This study investigates the solubility of several solutes, including KI, NaNO3, KNO3, HCl, NH4Cl, NH3, KCl, NaCl, KClO3, and SO2, in water as a function of temperature from 0°C to 150°C. The data presents solubility values (in grams of solute per 100 grams of water) demonstrating how temperature affects the dissolution of solids and gases. We explore the characteristics of saturated, supersaturated, and unsaturated solutions, providing insights into solution dynamics and practical applications in chemistry.

Solubility of Various Solutes in Water vs. Temperature
E N D
Presentation Transcript
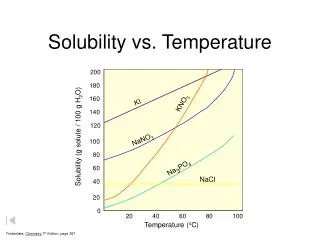
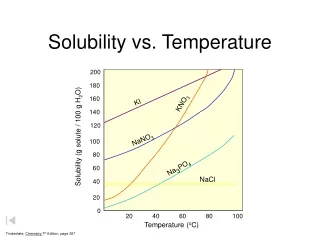
gases solids Solubility vs. Temperature 140 KI 130 120 NaNO3 110 100 KNO3 90 80 HCl NH4Cl 70 Solubility (grams of solute/100 g H2O) 60 NH3 50 KCl 40 30 NaCl KClO3 20 10 SO2 0 10 20 30 40 50 60 70 80 90 100 Temperature (oC)
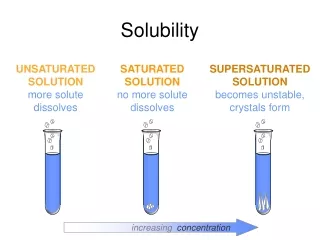
150 Saturated Supersaturated solution 100 Solubility(g/100 g H2O) Unsaturated solution 50 0 25 50 75 100 Temperature (oC)