Periodic Trends: Elements and Bonding
280 likes | 343 Views
Explore the properties of elements, such as reactivity, radius, electronegativity, and metallic character. Learn about different types of chemical bonds. Test your knowledge on ions and their classifications.

Periodic Trends: Elements and Bonding
E N D
Presentation Transcript
1. Which of the elements is the metalloid? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
2. Which of the elements would be the least reactive metal? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
3. Which of the elements would be the most reactive nonmetal? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
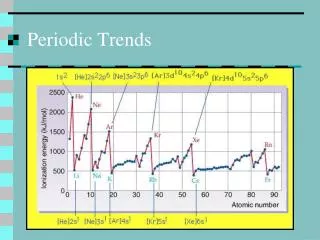
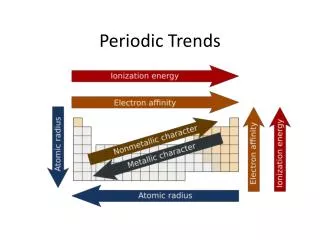
4. Which of the elements would have the largest radius? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
5. Which of the elements would have the highest electronegativity? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
6. Which of the elements would have the Lowest ionization energy? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
7. Which of the elements would have the greatest electron affinity (the most negative number)? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
8. Which of the elements would have the highest metallic character? • Aluminum • Silicon • Chlorine • Magnesium • Sulfur
9. Which of the following would have the larger radius? • Chlorine • Chlorine Ion
10. Which of the following would have the smaller radius? • Lithium • Lithium Ion
11. Which of the following would have the smaller radius? • Oxygen • Oxygen Ion
12. Which of the following would have the larger radius? • Sodium • Sodium Ion
13. Rubidium (n=37) would have all of the following characteristics except: • High Conductivity • High Malleability • High Brittleness • Solid at Room Temperature • 1 Valence Electron
14. Bromine (n=35) would have which of the following characteristics? • High Conductivity • High Malleability • High Brittleness • Solid at Room Temperature • 7 Valence Electrons
15. Which of the following would have the highest melting point? • Fluorine • Chlorine • Bromine • Iodine • Astatine
16. Which of the following would have the lowest density? • Fluorine • Chlorine • Bromine • Iodine • Astatine
17. Which of the following would have the highest density? • Potassium • Iron • Bromine • Krypton
18. Which of the following would have the lowest boiling point? • Potassium • Iron • Bromine • Krypton
19. Which of the following is isoelectronic to Na1+? • F1+ • Al3- • Mg1+ • Ne
20) What type of bond contains an active metal and a nonmetal? • metallic • covalent • ionic • hydrogen
21) What type of bond contains a nonmetal bonded with a nonmetal? • metallic • covalent • ionic • hydrogen
22) What type of bond contains a metalloid and a nonmetal? • metallic • covalent • ionic • hydrogen
23) What type of bond would form the alloy, pewter, with tin and lead? • metallic • covalent • ionic • hydrogen
24) N2 is a(n)? • anion • cation • diatomic molecule • polyatomic ion
25) N3- is a(n)? • anion • cation • diatomic molecule • polyatomic ion
26) NH4+ is a(n)? • anion • monatomic ion • diatomic molecule • polyatomic ion
27) Na+ is a(n)? • anion • cation • diatomic molecule • polyatomic ion