Periodic Trends
Periodic Trends. Mrs. Coyle Chemistry. Part I – Atomic Size. Atomic Radius. Measures as distance from nucleus to nucleus and divided by 2. Unit commonly used is pm picometer= 10 -12 m Example: iodine atomic radius 140pm. How does atomic radius change across a period?.

Periodic Trends
E N D
Presentation Transcript
Periodic Trends Mrs. Coyle Chemistry
Atomic Radius • Measures as distance from nucleus to nucleus and divided by 2. • Unit commonly used is pm • picometer= 10-12m • Example: iodine atomic radius 140pm
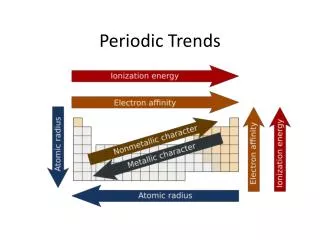
How does atomic radius change across a period? • It is smaller to the right. • Why? • More protons in the nucleus higher electrical force pulls electrons closer to nucleus.
How does atomic radius change down a group? • It is larger down the group. • Why? • Valence electrons are at higher energy levels and are not bound as tightly to the nucleus because they are screened or shielded ( pushed away) by other electrons in inner levels.
Note • There are some exceptions. Example column 13.
Example: Which is larger: a lithium atom or a fluorine atom? A lithium atom
Example: • Which is larger: an arsenic atom or a sulfur atom? • An arsenic atom
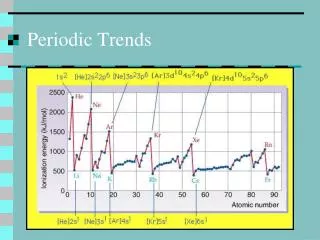
Ionization energy • Ionization energy is the amount of energy needed to remove an electron from a gaseous atom. • First ionization energy – 1+ • Second ionization energy – 2+
Ion • Positive ion ---removal of electron • Negative ion--- addition of electron
How does ionization energy change down a group? • The first ionization energy decreases as you move down a group. • Why? • The size of the atom increases. • Electron is further from the nucleus.
How does ionization energy change across a period? • The first ionization energy increases as you move from left to right across a period. • Why? • Nuclear charge increases while shielding is constant. • Attraction of the electron to the nucleus increases.
Ionic size • Metallic elements easily lose electrons. • Non-metals more readily gain electrons. How does losing or gaining an electron effect the size of the atom (ion) ?
Positive ions • Positive ions are always smaller that the neutral atom. Loss of outer shell electrons.
Negative Ions • Negative ions are always larger than the neutral atom. Gaining electrons.
Ion size trends in periods. • Going from left to right there is a decrease in size of positive ions. • Starting with group 5, there is sharp increase followed by a decrease in the size of the anion as you move from left to right.
Ion size trends in columns. • Ion size increases as you move down a column for both positive and negative ions
Electronegativity: the ability of an atom in a bond to pull on the electron. (Linus Pauling)
Electronegativity • When electrons are shared by two atoms a covalent bond is formed. • When the atoms are the same they pull on the electrons equally. Example, H-H. • When the atoms are different, the atoms pull on the electrons unevenly. Example, HCl
Trends in Electronegativity • Electronegativity generally decreases as you move down a group. • Electronegativity of the representativeelements (Group A elements) increases as you move across a period.
Electronegativities of Some Elements Element Pauling scale F 4.0 Cl 3.0 O 3.5 N 3.0 S 2.5 C 2.5 H 2.1 Na 0.9 Cs 0.7
Note • Most electronegative element is F (EN 4.0) • Least electronegative stable element is Cs (EN 0.7)
Summary Nuclear charge increases Shielding increases Atomic radius increases Ionic size increases Ionization energy decreases Electronegativity decreases Shielding is constant Atomic Radius decreases Ionization energy increases Electronegativity increases Nuclear charge increases