The Nucleus: A Chemist’s View
160 likes | 342 Views
The Nucleus: A Chemist’s View. Chapter 20 E-mail: benzene4president@gmail.com Web-site: http://clas.sa.ucsb.edu/staff/terri/. The Nucleus: A Chemist’s View. 1. Supply the missing particle for each of the following nuclear reactions: a. 73 Ga 73 Ge + ? b. 192 Pt 188 Os + ?

The Nucleus: A Chemist’s View
E N D
Presentation Transcript
The Nucleus: A Chemist’s View Chapter 20 E-mail: benzene4president@gmail.com Web-site: http://clas.sa.ucsb.edu/staff/terri/
The Nucleus: A Chemist’s View 1. Supply the missing particle for each of the following nuclear reactions: a. 73Ga 73Ge + ? b. 192 Pt 188Os + ? c. 205Bi 205Pb + ? d. 241Cm + ? 241Am
The Nucleus: A Chemist’s View 2. Write an equation for each of the following: a. Electron capture to 68Ga b. Positron emission of 62Cu c. Alpha decay of 212Fr d. Beta decay of 129Sb
The Nucleus: A Chemist’s View 3. The radioactive isotope 247Bk decays by an alpha and beta series ending in 207Pb. How many alpha and beta particles were emitted in the series?
The Nucleus: A Chemist’s View 4. The only stable isotope of fluorine is fluorine-19. Predict possible modes of decay for fluorine-21 and fluorine-18.
The Nucleus: A Chemist’s View 5. The first atomic explosion was detonated on July16, 1945. What fraction of the strontium-90 (t1/2 = 28.8 yr) will remain as of July 16, 2014?
The Nucleus: A Chemist’s View 6. The sun radiates 3.9 x 1023 J of energy into space every second. What is the rate at which mass is lost from the sun?
The Nucleus: A Chemist’s View 7. The most stable nucleus in terms of binding energy per nucleon is 56Fe. If the atomic mass of 56Fe is 55.9349 amu, calculate the binding energy per nucleon for 56Fe. (neutron = 1.67493 x 10-27 kg, proton = 1.67262 x 10-27 kg)
The Nucleus: A Chemist’s View 8. A positron and an electron annihilate each other upon colliding, thereby producing energy in the form of 2 photons. Calculate the wavelength of the light produced. (mass of an electron = 9.10938 x10-31kg)
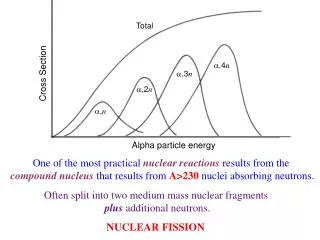
The Nucleus: A Chemist’s View 9. Define fusion and fission.
The Nucleus: A Chemist’s View 10. A freshly isolated sample of 90Y was found to have an activity of 9.8×105 disintegrations per minute at 1:00 pm on December 3, 2000. At 2:15 pm on December 17, 2000, its activity was redetermined and found to be 2.6×104 disintegrations per minute. Calculate the half-life of 90Y.