Electrolytic Cells Notes
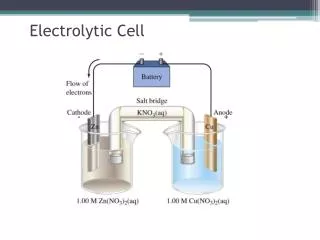
Electrolytic Cells Notes. Source or use of electricity. Electrode. Cell. –. Molten or aqueous chemicals. +. –. +. –. +. Overview. In “electrolytic cells”, electricity is used to force chemicals to undergo a redox reaction

Electrolytic Cells Notes
E N D
Presentation Transcript
Source or use of electricity Electrode Cell – Molten or aqueous chemicals + – + – + Overview • In “electrolytic cells”, electricity is used to force chemicals to undergo a redox reaction • In “galavanic cells”, electricity is produced spontaneously from a redox reaction “Cells” are containers of liquid with electrodes:
Voltaic vs. Electrolytic Cells • Voltaic • Spontaneous redox • Exothermic • Separate half cells • Salt bridge • Active electrodes • Positive cell potential • Generates electricity • Ex. battery • Electrolytic • Non-spontaneous redox • Endothermic • Same cell (one compartment) • No salt bridge • Inert electrodes • Negative cell potential • USES a battery source • Ex. Electroplating (Sn, Ag), electrolysis of water, separation of molten NaCl, recharging batteries
How can we use this? The stoichiometry of electrolysis processes can quantify “how much chemical change occurs with the flow of a given current for a specific time”.
Faraday’s Law • Faraday’s Law - The amount of substance undergoing chemical reaction at each electrode during electrolysis is directly proportional to the amount of electricity that passes through the electrolytic cell. • A faraday is the amount of electricity that reduces one equivalent of a species at the cathode and oxidizes one equivalent of a species at the anode. • 1 faraday of electricity ≡ 6.022×1023e
Coulomb A coulomb is the amount of charge that passes a given point when a current of one ampere (A) flows for one second. 1 ampere (amp) = 1 coulomb/second 1 faraday = 6.022 * 1023 electrons 1 faraday = 1 mole electrons 1.0 mole electrons = 96,485Coulombs
Sample Problem Example: Calculate the mass of palladium produced by the reduction of palladium (II) ions during the passage of 3.20 amperes of current through a solution of palladium (II) sulfate for 30.0 minutes.
Practice Ch 17: 77ab, 79 ab, 81* Electrochemistry Review Packet (with some more thermochemistry)