Atomic Theory
E N D
Presentation Transcript
Scientific Method Starts with a question:
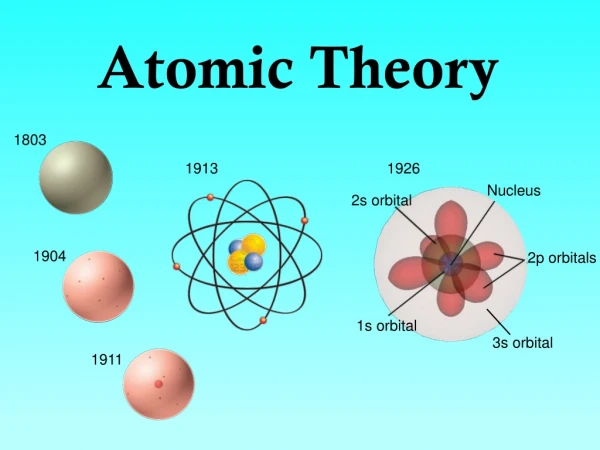
In the Beginning… 400 BC Democritus: Believed atoms were the smallest indivisible and indestructible forms of matter.
Aristotle: Disagreed. He thought all matter was continuous and part of the elements fire, earth, water, and air.
2000 years of darkness… 1690’s Scientific Experiments Law of conservation of mass- mass is not created nor destroyed
1808 Dalton’s Atomic Theory • Matter is made up of atoms • All atoms of an element are the same • Atoms cannot be subdivided, created, nor destroyed • Atoms combine in ratios to form compounds • In chemical reactions, atoms are combined, separated, or rearranged
Dalton’s Model- 1808 Tiny, indestructible particles with no internal structure
1890- J.J. Thomson What is the nature of these mysterious, invisible carriers of electricity?
Electrons are negative A cathode ray is deflected by a magnet. 4.2
Electrons have mass Later proved by Millikan in 1916: 9.11 x 10^-31kg
Summary In a sentence or two, describe the relationship between the cathode ray and electron.
Thomson- 1890Cathode Tubes • Electrons are negatively charged • Electrons have mass • Electrons behave like particles • Exist in all atoms Plum-Pudding Model
Unanswered Questions If electrons have such a small mass, what accounts for the higher mass? Atoms can’t be made solely out of electrons because atoms are neutral so something must balance it out.
Rutherford- 1911 • Mass concentrated in tiny core called nucleus • Electrons orbit the nucleus -If the nucleus is the size of marble, then the atom is the size of a football field Planetary Model
Homework Atomic Timeline Show how scientists views have changed over time. -date -person/event -pictures