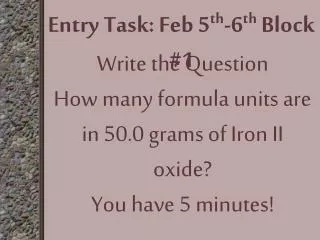Calculating Formula Units in Iron(II) Oxide: A Percent Composition Lesson
180 likes | 319 Views
In this task, we will determine how many formula units are present in 50.0 grams of Iron(II) oxide (FeO). We will explore the relationship between particles, moles, and grams, while also performing a self-check on these concepts. The session will include a discussion on percent composition, highlighting how to calculate the percentage of each element in a compound, with examples including magnesium oxide and iron(III) oxide. This will serve as a foundational skill in chemistry for understanding compound compositions.

Calculating Formula Units in Iron(II) Oxide: A Percent Composition Lesson
E N D
Presentation Transcript
Entry Task: Feb 5th-6thBlock #1 Write the Question How many formula units are in 50.0 grams of Iron II oxide? You have 5 minutes!
Agenda: Discuss Particles <-> Moles<-> Grams Self Check on Concepts Discuss Self-Check Notes on Percent Composition HW: Percent Composition ws
I can… • Calculate particles moles grams • Explain what is meant by the percent composition of a compound.
Discuss Particles Moles Grams Avervision for answers
Clear off your Desk!! Calculators Periodic Tables Needed
Discuss Self Check Avervision for answers
Percent Composition is the percent of each element in a compound. Percent composition is a step in determining the ratio in elements in a compound/molecule.
Example: Magnesium oxide- MgO By just looking at this compound, It LOOKS like it 50/50 of magnesium oxide. But according to its molar mass, it is not 50/50. Its molar mass is 24.305 g for Mg + 15.999 g for O, = 40.304 g 24.305g (Magnesium mass) 40.304 grams (mass of MgO) Magnesium = 0.60 X 100 = 60% Oxygen 15.999 g (oxygen mass) 40.304 grams (mass of MgO) = 0.40 X 100 = 40%
Iron III oxide Fe2O3 Fe = 55.845 x 2 = 111.69 g 159.687 g 0.6994 X 100 = 69.9% 159.687 g O = 15.999x 3 = 47.997g 0.3005 X 100 = 30.1% 159.687 g
Barium hydroxide Ba(OH)2 Ba= 137.33 g X 1 = 137.33 g 171.3438 g = 0.801 X 100 = 80.1% O= 15.999 g X 2 = 31.998 g 171.3438 g = 0.187 X 100 = 18.7% H= 1.0079 g X 2 = 2.0158 g 171.3438 g = 0.0117 X 100 = 1.2% 171.3438 g
Lead (II) nitrate Pb(NO3)2 Pb= 207.2 g X 1 = 207.2 g 331.208 g = 0.626 x100 = 62.6% N= 14.007 g X 2 = 28.014 g 331.208 g = 0.0845 x100 = 8.46% 331.208 g = 0.289 x100 = 28.9% O= 15.999 g X 6 = 95.994 g 331.208 g
Ammonium sulfate (NH4)2SO4 131.044 X 100 = 21.4 % N= 14.007 g X 2 = 28.014 g H= 1.0079 g X 8 = 8.06 g 131.044 X 100 = 6.2% S= 30.974 g X 1 = 30.974 g 131.044 X 100 = 23.6 % O= 15.999 g X 4 = 63.996 g 131.044 X 100 = 48.8.4 % add up % = 100% 1 mole = 131.044 grams
Hydronium nitride (H3O)3N 71.1 grams x 100= H= 1.0079 g X 9 = 9.071 g = 12.7 % O= 15.999 g X 3 = 47.997 g 71.1 grams x 100= = 67.6 % N= 14.007 g X 1 = 14.007 g 71.1 grams x 100= = 19.7 % 71.1 grams
Aluminum cyanide Al(CN)3 Al= 26.98 g X 1 = 26.98 g 105 grams x 100= = 25.6 % 105 grams x 100= = 34.3 % C= 12.0107 g X 3 = 36.032 g 105 grams x 100= = 40.0 % N= 14.007 g X 3 = 42.021 g 105 grams