Nuclear Power
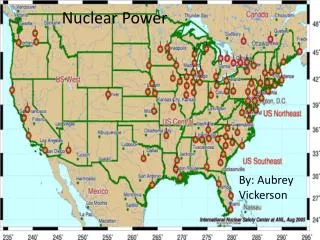
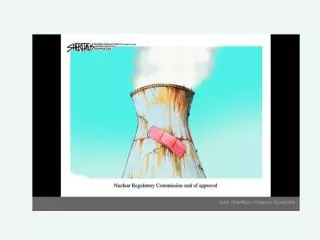
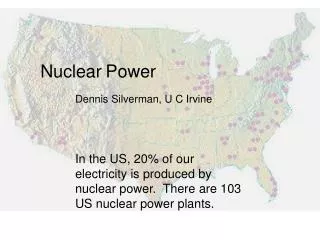
Nuclear Power . Locations of Nuclear Power plants in the US. Locations of Nuclear Power plants in the World. Do Nuclear Power plants Pollute?. No they don’t. This is Steam being released. . Nuclear Power Plant Operation. Uranium ore. Nuclear Reactor Fuel. Uranium ore is refined then

Nuclear Power
E N D
Presentation Transcript
Nuclear Reactor Fuel Uranium ore is refined then formed into pellets.
Nuclear Reactor Fuel These Pellets are then put into Fuel rods which are Assembled Into packs of Fuel Rod Assemblies
Parts Of an Atom • Protons • Neutrons • electrons
Protons Protons have a positive charge and are located in the nucleus of the atom.
Neutrons Neutrons are located in the nucleus and have no charge
Electrons • Electron are found on • The outside of the atom. • An electrically balanced • atom will have the same • number of electrons • and protons
What is Nuclear Decay? Nuclear decay is when the nucleus goes through a splitting process called nuclear Fission resulting in a different element(s) along with other products including ionizing radiation.
Ionizing Radiation • Ionizing radiation is produced by unstable atoms. Unstable atoms differ from stable atoms because they have an excess of energy or mass or both. • Unstable atoms are said to be radioactive. In order to reach stability, these atoms give off, or emit, the excess energy or mass. These emissions are called radiation.
4 types of ionizing Radiation • Alpha Helium Nucleus • Beta Electron • Gamma EM Radiation • Neutrons N0 These are other products that can be produced along with the new element
Ionizing Radiation alpha particle beta particle Radioactive Atom Neutron X-ray gamma ray
Alpha radiation • Nucleus of a helium atom • Symbolically represented: • Chemically written: 4He2 • Least Destructive Radiation • Can be stopped by a sheet of thick paper
Alpha Particles Alpha Particles: 2 neutrons and 2 protons They travel short distances, have large mass Only a hazard when inhaled
Beta radiation • Electron • Symbolically represented: • Chemically written: e- • More Destructive than Alpha Radiation
Beta Particles Beta Particles: Electrons or positrons having small mass and variable energy. Electrons form when a neutron transforms into a proton and an electron or:
Gamma radiation • High energy Electro-Magnetic Radiation • Symbolically represented: • Most Destructive Radiation • Very difficult to stop
Gamma Rays Gamma Rays (or photons): Result when the nucleus releases Energy, usually after an alpha, beta or positron transition
Neutron Radiation • High energy radiation • Symbolically written as n • Chemically written n0 • Is a result of fission and/or fusion • Often produced in particle accelerators • New Evidence suggests that Neutrinos (neutron radiation) can travel faster than light
Nuclear Half-Life Equation • Ni* (1/2)nt1/2 = Nf • Ni – Initial amount of radioactive material • nt1/2 -# of half-lives • Nf– Final amount of radioactive material To get nt1/2, you must divide time given in problem by the half-life.
Nuclear halflife examples • Polonium210 • Half Life: 138 days • Alpha decay • Strontium90 • Half Life: 28.5 years • Beta decay • Cobalt60 • Half Life: 5.27 years • Gamma decay
Alpha Decay Example • Polonium210 • Half Life: 138 days • Alpha decay If you have 48kg of Polonium 210, How much will be left after 138 days? Ans: 24 kg How much will be left after 276 days? (2 half lives) Ans: 12 kg How much will be left after 414 days? (3 half lives) Ans: 6 kg
Beta Decay Example • Strontium90 • Half Life: 28.5 years • Beta decay If you have 30kg of Strontium 90, How much will be left after 28.5 years? Ans: 15 kg How much will be left after 57 years? (2 half lives) Ans: 7.5 kg How much will be left after 85.5 years? (3 half lives) Ans: 3.75 kg
Gamma Decay Example • Cobalt60 • Half Life: 5.27 years • Gamma decay If you have 1 kg of Cobolt 60, How much will be left after 5.27 years? Ans: 0.5 kg How much will be left after 10.54 years? (2 half lives) Ans: 0.25 kg How much will be left after 15.81 years? (3 half lives) Ans: 0.125 kg