All about catalysts
170 likes | 570 Views
All about catalysts. Catalysts are substances that increase the rate of a chemical reaction by reducing the activation energy, but which is left unchanged by the reaction. (MODIFIED from URL)

All about catalysts
E N D
Presentation Transcript
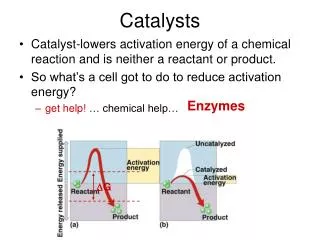
Catalysts are substances that increase the rate of a chemical reaction by reducing the activation energy, but which is left unchanged by the reaction. (MODIFIED from URL) This definition allows for the possibility that small amounts of the catalyst are lost in the reaction or that the catalytic activity is slowly lost. However, the catalyst affects only the rate of the reaction, it changes neither the thermodynamics of the reaction nor the equilibrium composition http://www.lboro.ac.uk/departments/cg/Projects/2002/dhonsi/Theory.html
Why are catalysts important? The principal purpose of catalysis is to increase the rate of the chemical reactions. It is important to understand the mechanism by which the reaction takes place. Catalysis is of crucial importance for the chemical industry: the number of catalysts applied in industry is very large and catalysts come in many different forms, from heterogeneous catalysts in the form of porous solids and homogeneous catalysts dissolved in the liquid reaction mixture to biological catalysts in the form of enzymes. A reaction catalysed by a heterogeneous catalyst can be represented by a flow chart
Making and breaking bonds absorb: adsorb? Reactant molecules are adsorbed at active sites onto the surface of the catalyst. This involves the formation of weak bonds between reactant molecules and the catalyst which causes other bonds in the reactant molecule to be stretched and weakened. The weakened structure is converted to another complex that is essentially the product attached to the catalyst. Finally, this complex breaks down to release the product molecule which moves away to leave the catalyst surface ready to interact with another reactant molecule
Mechanisms: Langmuir-Hinshelwood-Hougen-Watson (LHHW) mechanism [2] <reference.html>: (A) Two molecules adsorb onto the surface. (B) They diffuse across the surface and interact when they are close. (C) A molecule is formed which desorbs.
Eley-Rideal (E-R) mechanism A) Molecules adsorbs onto the surface. (B) Another atom passes by which interacts with the one on the surface. (C) A molecule is formed which desorbs.
http://www.saskschools.ca/curr_content/chem30_05/graphics/2_graphics/catalyst1.gifhttp://www.saskschools.ca/curr_content/chem30_05/graphics/2_graphics/catalyst1.gif
Intermediate Compound theory Catalyst = C A + B -------------> D slow reaction C A + B -------------> D ? A + C -------------> [AC] intermediate produced quickly [AC] + B-------------> D + C produced quickly
The characteristic double humped profile for an intermediate compound theory. Notice again the lower EA A+B [AC] D modified image! http://www.docbrown.info/page07/SSquestions/catprofile.gif
The active site on the enzyme attaches to a substrate molecule (such as a disaccharide) forming an enzyme-substrate complex. While attached to the substrate, the enzyme causes a weakening of certain chemical bonds in the substrate molecule, resulting in a breakdown (hydrolysis) of the substrate into two smaller product molecules (such as two monosaccharides). The enzyme is unaltered during the reaction and is free to catalyze the breakdown of another substrate molecule. If the active site on the enzyme is blocked by a poison molecule, this vital hydrolysis reaction cannot occur. See the next section
Active Site Of Enzyme Blocked By Poison Molecule In this illustration a poison molecule has bonded to the active site of an enzyme. Now the enzyme is unable to recognize the substrate molecule that it normally conjugates with because its active site has been blocked. In locoweed poisoning of livestock, the alkaloid swainsonine blocks a key enzyme called mannosidase. When the active site of mannosidase is blocked, it cannot catalyze the breakdown of the sugar mannose, resulting in a lethal accumulation of mannose in vacuoles of cells of the central nervous system (brain and spinal cord). The actual vacuoles are swollen organelles called lysosomes where the enzymatic breakdown process normally occurs. The afflicted animal becomes paralyzed and eventually dies. A similar scenario occurs in infants suffering from the storage disease mannosidosis. In this disease, the vital enzyme mannosidase is lacking due to a mutant recessive gene. At the present time there is no cure for this disease. It is passed on via heterozygous carriers, and shows up with a 25 percent probability when two heterozygous individuals have a child. One of the better known storage diseases is Tay Sachs Disease, in which nerve cells fill up with a lipid called ganglioside or GM2 because they lack the vital enzyme HEX A needed to break down GM2
http://fajerpc.magnet.fsu.edu/Education/2010/Lectures/17_Enzymes_files/image012.jpghttp://fajerpc.magnet.fsu.edu/Education/2010/Lectures/17_Enzymes_files/image012.jpg