Scenario
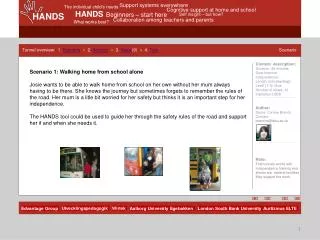
Scenario. The interior of the tank car was washed out & cleaned with steam. Then all the outlet valves were shut and the tank car was sealed. All the workers went home for the evening and when they returned, this is what they found. Scenario. Driving Question.


Scenario
E N D
Presentation Transcript
Scenario • The interior of the tank car was washed out & cleaned with steam. Then all the outlet valves were shut and the tank car was sealed. All the workers went home for the evening and when they returned, this is what they found.
Driving Question What was happening inside the tanker and outside the tanker that made it crush? Individual Brainstorm • WHY did the tanker crush? • HOW did the tanker crush? Think about “before”, “during” and “after”, and draw a diagram using the images on your sheet.
Group Poster • With your group share your ideas about how/why the tanker imploded. • Come up with a group model of what happened to the tanker. • Make sure to include drawings of the molecules to show what was happening inside or outside of the tanker. • Recorder 1 – Draws the picture of tanker and groups ideas about why it’s imploding • Recorder 2 – Uses words to explain groups ideas. • Reporter – Reports group’s ideas to the class.
Bellringer • How is the pop can similar to the crushing tanker? How are they different?
Common representation • Pressure Larger Equal Smaller • Should pressure be thought of as a push or a pulling force? • If pressure is a pushing force- is it correct to say…..”the air was sucked out of the can”. Pressure: Pressure is a measure of the force (pushing force) applied over a unit area.
Common representation • Temperature • Temperature- speed of molecules.
Common representation • Pressure Larger Equal Smaller • Temperature Hotter Room Temp. Colder Keep these in mind: • Pressure inside can vs. pressure outside can • Temperature changes- speed of molecular • Temperature changes- phase of matter (liquid or gas)
What might affect how much the can crushes? • I think _____________________ will cause the can to ______________. Experiment 1: Experiment 2: Experiment 3: Experiment 4: Experiment 5: Experiment 6:
Explain Results • In your explanation of your experimental results make sure to include: • Movement of molecules (speed) • Phase of matter (when is the water a gas, when it is it becoming liquid?) • What is causing pressure inside can? • How is the pressure outside can different from inside? Where do I want the answer to these questions?
How a thermometer works: • Molecules from the warm liquid crash into the glass of the thermometer and transfer energy to the alcohol in the thermometer. • The energy makes the molecules move faster in the thermometer • The alcohol molecules spread out and expand noted by the alcohol rising in the tube.
When a sample of gas, liquid or solid are heated its thermal energy increases. • Gas pressure is a measure of the collisions of the molecules with the walls of the container.
How a barometer works: • When the atmospheric pressure increases it pushes mercury in the barometer up higher. • When the atmospheric pressure decreases the column of mercury decreases in height.
Charles's Law (cont.) • Charles’s law states that if pressure is held constant, the volume of a given amount of gas is directly proportional to its kelvin temperature. • If volume goes up so does temperature. Note: Zero on the Celcius scale was chosen for convenience, zero on the Kelvin scale relates to particle motion.
Boyle's Law • Boyle’s law states that if temperature is held constant, the volume of a fixed amount of gas varies inversely with the pressure.
Gay-Lussac's Law • Gay-Lussac’s law states that if volume is held constant, the pressure of a fixed amount of gas varies directly with the kelvin temperature.
Bell Jar Demo • We now know all of the relationships possible between pressure, volume and temperature. • Which relationship describes what is happening with the bell jar demos? • Temperature and Volume • Pressure and Volume • Pressure and Temperature
Pressure and Particles • Pressure and the number of particles have a direct relationship. • As we increase the number of particles in a space, we also increase the pressure inside the space.
Relationship Application • If the pressure doubles what happens to volume? • The volume will halve. • If the temperature doubles what happens to the pressure? • The pressure will double. • If the temperature triples what will happen to the volume? • The volume will triple.
Unit Review • A common theme in chemistry is the importance of units. Units are useful for measuring values, and although the units we use may change, the values of what they measure remain constant. • The gas laws deal mainly with the units of Temperature, Pressure, and Volume. • Make a 2-T chart in your notebook, a column each for pressure, temperature, and volume label it: Units.
Gas Laws • The following are all units that you may be using over the rest of the year. For each variable, add it to the correct column in your chart. • It is YOUR responsibility to recognize these units. • ˚C • atm • L • mL • torr • ˚F • mm Hg • bar • cm3 • K • psi • m3
To use the combined gas law correctly you must convert degrees Celsius to Kelvin.
Practice Makes Perfect Practice Problem 1: The temperature inside my refrigerator is about 40 Celsius. If I place a balloon in my fridge that initially has a temperature of 220 C and a volume of 0.5 liters, what will be the volume of the balloon when it is fully cooled by my refrigerator?
Practice Makes Perfect Practice Problem 2: Synthetic diamonds can be manufactured at pressures of 6.00 x 104 atm. If we took 2.00 liters of gas at 1.00 atm and compressed it to a pressure of 6.00 x 104 atm, what would the volume of that gas be?
Practice Makes Perfect Practice Problem 3: If I initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 K, and then I raise the pressure to 14 atm and increase the temperature to 300 K, what is the new volume of the gas?
Practice Makes Perfect Practice Problem 4: A container of gas is initially at 0.500 atm, 13 ˚C, and 2L. What will the pressure be if the volume increase to 3 L and has a temperature of 105 ˚C?
Practice Makes Perfect A container of gas is initially at 0.500 atm, 25.0oC, and 2.00L. What will the pressure be if the volume increase to 3.00 L and has a temperature of 125oC? (Use Sig Figs!) • 0.445 atm • 0.00000281 atm • 761 atm • 0.148 atm
Gas laws A gas has a volume 12.3 L and a pressure of 4.00 atm. If the pressure increases to 6.00 atm, what is the final volume. (Sig Figs!) • 8.2 L • 18.45 L • 8.20 L • 18.5 L
Practice Makes Perfect Synthetic diamonds can be manufactured at pressures of 6.00 x 104 atm. If we took 2.00 liters of gas at 1.00 atm and compressed it to a pressure of 6.00 x 104 atm, what would the volume of that gas be? (Use Sig Figs!) • 120,000 L • 0.00003 L • 0.0000333 L • 0.0333 mL
Assessment Atoms are in their lowest energy state at what temperature? • 0° Celsius • 0° Fahrenheit • –100° Celsius • 0 kelvin • A • B • C • D
Practice Makes Perfect The temperature inside my refrigerator is about 4o Celsius. If I place a balloon in my fridge that initially has a temperature of 22o C and a volume of 0.5 liters, what will be the volume of the balloon when it is fully cooled by my refrigerator? (Show 4 Sig Figs!) • 0.0909 L • 0.4695 L • 0.09090 L • 0.46940 L
Practice Makes Perfect If I initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 K, and then I raise the pressure to 14 atm and increase the temperature to 300 K, what is the new volume of the gas? (Be accurate to the thousandths place.) • 29.572 L • 0.064 L • 0.148 L • 0.025 L
Gas Laws • Many technological advancements have come from our understanding of the gas laws. • Our cars rely on the relationship between temperature and volume. • Hot air balloons also rely on the relationship between temperature and volume. • Similarly, air conditioners and refrigerators utilize the relationship between pressure and temperature.
Gas Laws • As a bunny dances with a porcupine, 12.3 L of nonsense is written on a slide having something to do with 4 atm. When Tanner and Kyle accidentally get attacked by the porcupine because they all are trying to ask the bunny to prom. The bunny ran away feeling scared to 6 atm. Now the boys aren’t sure what they are solving for.
Gas laws • A gas has a volume 12.3L and a pressure of 4 atm. If the pressure increases to 6 atm, what is the final volume.