Enzymes
190 likes | 352 Views
Enzymes. http:// www.bing.com/videos/search?q=enzyme+action&view=detail&mid=266902AB5627FB3EE9CC266902AB5627FB3EE9CC&first=0&FORM=LKVR. A ________ reaction is a process that changes one set of chemicals into another set of chemicals. .

Enzymes
E N D
Presentation Transcript
http://www.bing.com/videos/search?q=enzyme+action&view=detail&mid=266902AB5627FB3EE9CC266902AB5627FB3EE9CC&first=0&FORM=LKVRhttp://www.bing.com/videos/search?q=enzyme+action&view=detail&mid=266902AB5627FB3EE9CC266902AB5627FB3EE9CC&first=0&FORM=LKVR
A ________ reaction is a process that changes one set of chemicals into another set of chemicals. Some chemical reactions occur ________, such as the combination of iron and oxygen to form an iron oxide called rust. Other reactions occur _________. When hydrogen gas is ignited in the presence of oxygen, the reaction is rapid and explosive. The elements or compounds that enter into a chemical reaction are known as ___________. The resulting material produced by that reaction are known as _______. 2H2 + O2 2H2O slowly chemical products quickly reactants
Some chemical reactions ________ energy & other chemical reactions ________ energy. What significance do these energy changes have for living things? In order to stay alive, organisms need to carry out reactions that require __________. Thus, every organism must have a source of energy to carry out chemical reactions. ________ get their energy by trapping and storing the energy from sunlight in energy-rich compounds. _________ get their energy when they consume plants or other animals. • Chemists call the energy that is needed to get a reaction started the _____________________. release energy plants absorb activation energy animals
Some chemical reactions that make life possible are too _______, or have activation energies that are too ______ to make them practical for living tissue. A __________ is a substance that speeds up the rate of a chemical reaction. Catalysts work by _________ a reaction's activation energy. _____________ are known as catalysts. lowering slow catalyst high ENZYMES
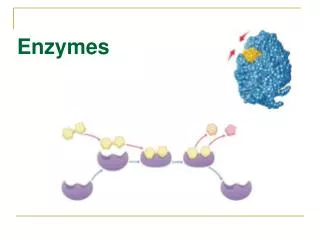
KEY CONCEPTS ABOUT ENZYMES ENZYMES are a type of _________________ Enzymes work like a ___________________ - the substrate is the lock and the enzyme is the key _____________ - they only work with one particular substrate (remember a key only fits one lock) ______________ - can be used over and over (the same key can fit a lock again and again) Affected by __________________ - can denature at high levels (reason why high fever can be life- threatening; the key gets bent and won't work in the lock any longer) Affected by ________-can denature at extreme pH ______________ - speeds up a chemical reaction (the quickest way to get in your house is by using the lock & key - not the only way, but the quickest way) reusable specific temperature protein pH lock & key catalyst
And now…. time for a review quiz
ENZYMATIC ACTION ENZYME PRODUCT SUBSTRATE
Catalysts _ _ _ _ _ up chemical reactions by _ _ _ _ _ _ _ _ the activation energy that gets the reaction going
MATCH THE LETTERS (A-F) TO THESE TERMS: Reaction pathway with enzyme Activation energy with enzyme Products Reactants Reaction pathway without enzyme Activation energy without enzyme
_ _ _ _ _ _ _ _ Not used up in a chemical reaction. Eventually, like most proteins, will slowly ‘wear out’ and be eliminated by the body. New ones are quickly manufactured by the cell(s).
_ _ _ _ _ _ _ _ 1:1 relationship between type of enzyme & type of substrate
Affected by high _ _ _ _ _ _ _ _ _ _ _
The ‘white’ of a chicken egg is pure protein (albumin). The yolk is mostly fat and cholesterol. Temperatures greatly higher than the normal body environment can cause proteins to begin to ‘denature’. This is why a high fever can cause problems with digestion and thought processes. On the other hand, many bacteria are even more sensitive to higher temperatures. This is why a fever kills them when you’re sick.
Affected By Extreme __ __ ACIDIC 0.0 7.0 14.0 NEUTRAL Average human pH ~ 7.2-7.4 BASIC