ECDCICA - CYCLES
E N D
Presentation Transcript
ECDCICA - CYCLES MATTER MUST CYCLE
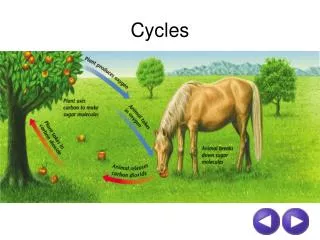
Cycles of Matter • Energy and matter move through the biosphere very differently. Unlike the one-way flow of energy, matter is recycled within and between ecosystems. The natural resources, water and nutrients, of the Earth are limited. Matter, including water and nutrients, moves through organisms and the biosphere through cycles.
Matter must be used over and over again by all living organisms. Nutrients are chemical substances that organisms need to survive. Every living thing needs nutrients to build tissues and carry out essential life functions. Matter in the form of waste or decay is returned to the land, water or atmosphere to be used over and over again.
1. Which elements are the basic building materials of life? • CHOPSN • Carbon, Hydrogen, Oxygen, Phosphorus, Sulfur, Nitrogen • 2. How does matter move among the biotic and abiotic parts of an ecosystem? • It cycles
Water Cycle • All living things need water to survive. Human blood is ~83% water, which helps digest our food, transport waste, and control body temperature. There is continuous movement of water on, above, and below the surface of the Earth. Water cycles between the ocean, atmosphere, land, surface water and living things.
About 90% of the water rainforest plants take in is released back into the ecosystem by evapotranspiration. The cool rainforest allows the water to condense and gives the plants a constant source of H20.
Heating of surface water to water vapor • Evaporation • Cooling of water vapor within the clouds • Condensation • The release of water from the clouds • Precipitation • Release of water vapor from plants • Evapotranspiration
The movement of water into the ground • Infiltration • The movement of water within the ground • Ground Water • The movement of water on the surface of the ground • Runoff • Deepundergroundwater storage that can supply wells and springs • Aquifer
1. Water can enter the atmosphere by which of the above processes? • Evaporation • Evapotranspiration
Carbon Cycle • Carbon compounds are the basis for the molecules necessary for life, including lipids (oils, fats, and waxes), proteins, nucleic acids (DNA, RNA), and carbohydrates. Carbon dioxide in the atmosphere provides carbon for living things, delivering it to animals via plants. Plants, bacteria and protists that are photosynthetic can capture carbon from the atmosphere and convert it to carbon compounds such as glucose. The three primary ways that carbon cycles in the abiotic and biotic parts of our biosphere.
I. Photosynthesis/Cellular Respiration in the Food Web • Carbon is picked up by plants from Carbon Dioxide, CO2, in the atmosphere during photosynthesis. Carbon from Carbon Dioxide, CO2, is used to make food C6H12O6 (sugar). Photosynthesis 6CO2+ 12H2O + Sunlight energy C6H12O6 + 6O2 + 6H2O
I. Photosynthesis/Cellular Respiration in the Food Web • When plants are eaten by consumers, the carbon is transferred and used in the consumer’s body. The carbon in the sugar/glucosecompound C6H12O6 is released back to the atmosphere as carbon dioxide, CO2, during cellular respiration.
I. Photosynthesis/Cellular Respiration in the Food Web • Cellular respiration converts chemical bond energy of C6H12O6, (sugar) to usable energy for the cell. Cellular Respiration occurs in plants and animals. Cellular Respiration C6H12O6 + 6O2 + 2ATP 6CO2 + 6H2O + 38 ATP (usable energy) (eventually heat loss)
I. Photosynthesis/Cellular Respiration in the Food Web • When plants and animals die, the carbon compounds are broken down by decomposers such as bacteria and fungi, releasing the carbon dioxideback into the land, atmosphere or water.
II. Combustion • Burial of dead organisms and conversion under pressure store carbon as coal, oil, or natural gas. Carbon is also stored as wood. • Duringcombustionwhen the material is burned as a fuel it releasesenergyand CO2.
III. Erosion • Erosionof calcium carbonate in limestone rock, animal skeletons, and shells of marine organisms returns carbon back to the oceans, where it is stored and later can be returned to the atmosphere as CO2.
Carbon Questions 1. Which processes releases carbon back into the atmosphere? Cellular Respiration, decomposition, erosion, volcanism, combustion of fossil fuels, burning forests 2. Why is it important that the ocean is a carbon sink or storage system? To help decrease global warming 3. Name 2 abiotic components and 2 biotic components of the carbon cycle. Abiotic- Atmosphere, water, soil and Biotic - plant, animal, decomposer
4. Producers/Plants use carbon dioxide during what process? Photosynthesis 5. Plants and animals release carbon dioxide during what process? CellularRespiration 6. What might happen to a forest if the trees were all cut down? Less O2, soil erosion, loss of nutrients, loss of organisms, change ecosystem
Oxygen Cycle • Oxygen makes up 90% of water (by mass), about 47% of the earth's crust, as well as 21% of dry air and makes up 65% of the human body (H20 by mass). Oxygen is cycled in our biosphere during the following processes:
Photosynthesis/Cellular Respiration in the Food Web • During the process of photosynthesis, water, H2O, is split into hydrogen and oxygen by the producer or plant. Oxygen gas, O2 is released into the atmosphere or aquaticecosystem. Photosynthesis 6CO2+ 6H2O + Sunlight energy C6H12O6 + 6O2
I. Photosynthesis/Cellular Respiration in the Food Web • Plants and animals on land or in water use oxygen for cellularrespiration. The oxygen in the atmosphere combines with hydrogen to form water, H2O, which is released during cellular respiration. Cellular Respiration C6H12O6+ 6O2 6CO2 + 6H2O + ATP (usable energy) ( eventually heat loss)
Oxygen is also needed for decomposition of organic material or waste.
II. Photolysis • Photolysis is the process where ultraviolet radiation breaks down water vaporinto oxygen gas.
III. Weathering • Weathering and erosion of exposed rock releases oxygen into the atmosphere or aquatic ecosystem. (ie. limestone rock, CaCO3 or calcium carbonate in shells). Plants and animals receive oxygen in this process.
IV. Water Movement • Oxygen in water is known as dissolved oxygen or DO. In nature, oxygen enters water when water runs over rocks and creates tremendous amounts of surface area.
Oxygen Questions 1. Plants release ________________during photosynthesis. oxygen, O2 2. Plants, Animals, and Decomposers use ___________ for cellular respiration. oxygen, O2 3. What four processes releases oxygen into the atmosphere, land or water? Photosynthesis, Weathering and Erosion, Water movement (Dissolved Oxygen), Photolysis
Nitrogen Cycle • All organisms require nitrogen to make amino acids which in turn are used to build proteins. Nitrogen is also needed for nucleic acids (DNA, RNA). In the form of nitrogen gas, N2, nitrogen is 78% of the atmosphere.
N2 must be converted to ammonia or nitrates by eitherlightning, industrial fixation (factories), burning transportation fuels or by bacteria in the soil before plants can use it.
I. Nitrogen Fixation • One way for plants to use nitrogen is for it to be converted to ammonia by nitrogen-fixing bacteria. The bacteria are often found living on the rootnodules of legume plants. The bacteria change the nitrogen (N2) in the atmosphere, to ammonia (NH3) during nitrogen fixation.
Plants take up the ammonia from the soil through their roots by assimilation (absorption). Plants use the nitrates to make amino acidswhich make proteins.
Animals get their nitrogen from eating plants or other animals to make amino acids the building blocks of proteins.
II. Nitrification • A second way for plants to use nitrogen is from animal waste or decaying plants and animals. The matter is broken down by decomposers and then the nitrite bacteriaconvert ammonia into nitrates (NO3) during nitrification.
The nitrates can be used by plants to make amino acidswhich make proteins, the most common organic compound in living things. • Animals get their nitrogen from eating plants or other animals to make amino acids the building blocks of proteins.
III. Denitrification • The nitrates in the soil or from an organism’s body can be converted by denitrifying bacteriainto nitrogen gas during denitrification. • The N2 is returned to the atmosphere so it can cycle over and over again.
Nitrogen Questions 1. What types of bacteria are part of the nitrogen cycle? nitrogen-fixing bacteria, nitrite bacteria, & denitrifying bacteria 2. What is nitrogen fixation by bacteria? Process by which bacteria on root nodules convert nitrogen into ammonia 3. What is nitrification? Process of waste or decayed organisms converted into nitrates. 4. What role does denitrification play in the nitrogen cycle? Process releases nitrogen back into the atmosphere
5. Besides bacteria what else can fix nitrogen (N2) into a usable form? Lightning, Industrial Fixation 6.Plants use ammonia or nitrates as a source of nitrogen to make amino acids the building blocks of _______________ and DNA and RNA. proteins 7. Animals use nitrogen from _______________ to make proteins. plants
Phosphorus Cycle • Phosphorus is an essential component in many molecules, such as ATP (energy molecule), phospholipids, RNA and DNA. Phosphorus can be a limiting factor because it is NOT available from the atmosphere, as carbon and nitrogen are. • Phosphorus does not exist as a gas and is primarily found in the solid form on land in soil, rocks and in ocean sediments.
I. Uplift/Weathering/Erosion • When rocks and sediments uplift, weathering and erosion gradually releases phosphorus as phosphate ions which are easily dissolved in water. Plants can absorb phosphate ions through their roots. • The rivers can carry phosphate to the oceans where marine plants can also absorb phosphate ions.
Plants convert the phosphorus into molecules, which are then passed through the ecosystem’s food chain. • Animals receive phosphate from eating plants or from consumers that ate plants.
II. Decomposition • Phosphorus is returned to the soil or ocean sediments by excretionsor death of plants and animals. • Decomposers break down the organism and return the phosphorus to the soil or ocean sediments.
Phosphorus Questions • Where is most of the phosphorus stored in an ecosystem? rocks/sediment 2. What state of matter is phosphorus NOT found? gas