Ionic Bonding
Ionic Bonding. Recall some information…. The number of valence electrons = group number for an element. Elements in the same group have similar chemical properties. Valence electrons are largely responsible for giving an element its chemical properties.

Ionic Bonding
E N D
Presentation Transcript
Recall some information… • The number of valence electrons = group number for an element. • Elements in the same group have similar chemical properties. • Valence electrons are largely responsible for giving an element its chemical properties. • This is because they are the only electrons that are responsible for forming bonds. Lewis Dot Structures can show us how many valence electrons an element contains. Notice, all Group 1A elements have one valence electron.
The Octet Rule When atoms form compounds they achieve the electron configuration of a noble gas.
Why do atoms form bonds? • All atoms are attempting to fill their outermost shell. • This makes them stable. “Happy” (Octet Rule) The bonds of friendship are hard to break Unfulfilled Life Lonely Electron Befriends Sad Halogen 7 8
Ions Formed when an atom gains or loses electrons; the atom will take on a charge (+ or -).
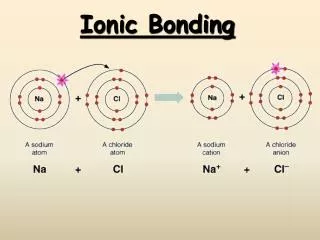
How are cations formed? • Metals will tend to lose electrons. • This will give them a positive charge. • Remember, losing negative things will make you more positive. • Na+, Mg2+, Al3+ • When naming cations, the name remains the same. Sodium has one electron in its outermost shell 1. Gain 7 electrons 2. Lose 1 electron +1 Sodium now has a complete outer shell; but now has one fewer electron than before! Sodium is now an ion and has a +1 charge.
Magnesium has 2 valence electrons; so it needs to get rid of both of them. Magnesium now has two more protons than electrons, giving it a positive charge. +2 +3 Aluminum is in group 3; therefore it needs to lose 3 valence electrons. Aluminum now has three more protons than electrons, giving it a positive charge.
How are anions formed? • Non-metals will gain electrons. • This will give them a negative charge. • Remember, gaining negative things will make you more negative. • F-, O2-, N3- • When naming the ion, the ending changes to –ide. Fluorine has 7 electrons in its outermost shell 1. Gain 1 electron 2. Lose 7 electron -1 Fluorine is now an ion and has a -1 charge. Fluorine now has a complete outer shell; but now has one more electron than before!
-2 Oxygen now has 2 more electrons than protons, giving it a negative charge. Oxygen has 6 valence electrons; it needs to gain 2. -3 Nitrogen has 5 valence electrons; it needs to gain 3. Nitrogen now has 3 more electrons than protons, giving it a negative charge.
Can you predict charges? • Yes! You do not have to memorize the charges of each element. • Since elements in the same group have the same number of valence electrons • All metals in the same group lose the same number of valence electrons • All non-metals in the same group gain the same number of valence electrons. • All elements in the same group will have the same charge. • Note: The transition metals do NOT follow this rule. Review your Ions POGIL packet for more information!
All Group 7A elements are gaining 1 electron (remember to get to 8); they will ALL have a -1 charge. All Group 6A elements are gaining 2 electrons; they will ALL have a -2 charge. All Group 1A elements are losing their only valence electron; they will ALL have a +1 charge. All Group 2A elements are losing both of their valence electrons; they will ALL have a +2 charge. +1 +2 -3 -2 -1 +3
Ionic Compounds Compounds composed of cations and anions; electrically neutral (even though ions have charges); properties of the compounds can be very different from the properties of the ions.
What is an example of an ionic compound? • Sodium Chloride (NaCl) – table salt! • Sodium is a highly reactive metal. • Chlorine is a poisonous gas. • Together they form a harmless (and necessary) compound.
How do ionic bonds form? • Bonds where electrons are transferred. • Occurs between cations and anions; metals and non-metals. This group 1A element needs to lose 1 electron; it transfers it to the group 7A element, which needs to gain 1 electron. Both atoms are now happy with full octets! Note: This slide uses Lewis Dot Structures
This cation has a +2 charge. The anions have -1 charges. This means you need two anions to bond with the cation. 1(2) + 2(-1) = 0 This cation has a +2 charge. The anion has a -2 charge. This you only need one of each to form the ionic bond. 1(2) + 1(-2) = 0 #cation(charge) + #anion(charge) = 0
What are the properties of ionic bonds? • High melting points – high boiling points. • Solid at room temperature. • They can conduct electricity when dissolved in water (H2O). • Tend to be hard and brittle. • They form crystals – 3D patterns of alternating cations and anions (crystal lattice). Review the PHET (Sugar and Salt Solutions) simulation for more information!
How do you write a Lewis Dot Structure for Ionic Compounds? • 1. Identify the elements in the formula. • Ex. Barium chloride -> Ba & Cl • 2. Write each element in its ionic form. • Ba+2 & Cl-1 (remember, the charges can be found by looking at the group number).
How do you write a Lewis Dot Structure for Ionic Compounds? • 3. Use the cross-over rule to determine the chemical formula of the compound. Formula: Ba1Cl2 +2 -1 Ba Cl
How do you write a Lewis Dot Structure for Ionic Compounds? • 4. Draw every atom’s Lewis Dot Structure in its neutral form. (remember, there are 2 Cl atoms). • 5. Draw arrows from the cation to the anion, indicating where the electrons will be pushed. For more examples, head over to the videos section on mrgarcia.netand watch the Ionic Bonding examples. This is what I would expect you to do on an exam, I must see the arrows for you to receive credit!