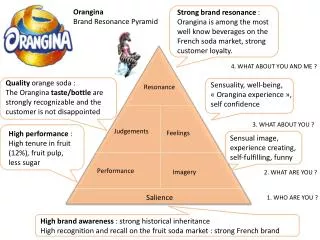
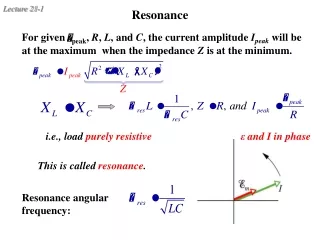
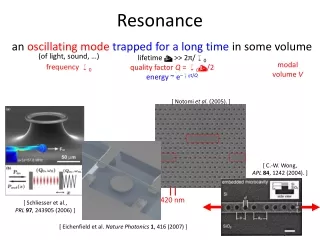
Resonance
Resonance. Example : O 3 Experimental evidence shows each of the oxygen-oxygen bonds are identical . There are certain cases in which a single Lewis structure cannot correctly describe a molecule. These molecules exhibit a phenomena known as resonance .

Resonance
E N D
Presentation Transcript
Resonance • Example: O3 • Experimental evidence shows each of the oxygen-oxygen bonds are identical. • There are certain cases in which a single Lewis structure cannot correctly describe a molecule. • These molecules exhibit a phenomena known as resonance. • Actual structure is an averageof the resonance structures.
Polyatomic Ions • Group of covalently bonded atoms that has a charge. (a charged molecule) • Example: NO3- (nitrate ion)
Molecular Geometry and VSEPR Theory • Whenever the atoms of a molecule bond together, the molecule takes on a particular shape. • Molecular geometry refers to the three-dimensional arrangement of a molecule’s atoms in space. • VSEPR theory is the model used to explain the shapes of molecules.
VSEPR – Valence Shell Electron Pair Repulsion Theory. • The main idea of this theory is that electron pairs on the central atom of a molecule will position themselves in such a way as to minimize repulsion. • There are two types of electron pairs: • bonding pairs • nonbonding pairs
Example – CH4 • 4 bonding / 0 nonbonding pairs • molecular geometry = tetrahedral • bond angles are 109.5° • Example – NH3 • 3 bonding / 1 nonbonding • molecular geometry = trigonalpyramidal • bond angles are 107° • Why are bond angles less?
Example – H2O • 2 bonding / 2 nonbonding • Molecular Geometry = bent • Bond Angle = 104.5°
VSEPR and Multiple Covalent Bonds • For the VSEPR model, multiple bonds are counted as one bonding pair when determine the shape of the molecule. • Example – H2CO3 • 3 bonding / 0 nonbonding • Molecular Geometry = trigonal planar • Bond Angles = 120°
Example – CO2 • 2 bonding / 0 nonbonding • Molecular Geometry = linear • Bond Angles = 180° • If a molecule exhibits resonance, any one of the resonance structures can be used to predict the shape. • Example – O3 • 2 bonding / 1nonbonding • Molecular Geometry = bent • Bond Angles = approx. 120°