Alkenes and bromine
160 likes | 553 Views
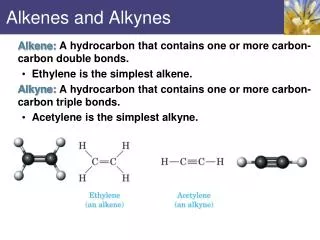
Alkenes and bromine. Starter. Attempt the reaction mechanism sheet. Learning outcomes. • Describe how heterolytic fission leads to electrophilic addition in alkenes, using bromine as an example. • Recall the reactions of alkenes. Addition of the hydrogen halides.

Alkenes and bromine
E N D
Presentation Transcript
Starter • Attempt the reaction mechanism sheet
Learning outcomes • Describe how heterolytic fission leads to electrophilic addition in alkenes, using bromine as an example. • Recall the reactions of alkenes.
Addition of the hydrogen halides • Write a brief sep-by-step method for the addition reaction of the hydrogen halides. • Remember to use key terms like... • Electronegativity • Dipole • πbond • Double bond • Heterolytic fission
Addition reaction of bromine • Bromine is a non-polar molecule and yet it is used as a test for unsaturation because it reacts with alkenes. • Using what you know about alkenes, bromine and electrons, see if you can come up with a reaction mechanism.
Prompts • Bromine is non-polar. Is there a way that a dipole could be induced in the bromine molecule? • Why will the double bond break? • What is the first bond to form? What are the two unstable products?
Bromine is non-polar but when it approaches a double bond, the electrons in the π-bond repel the electrons in the Br-Br bond. This inducesa dipole in the Br2 molecule. • The Br2 molecule is now slightly polar. • The electron pair in the π-bond is now attracted to the slightly positive bromine atom, causing the double bond to break. • A new bond forms between a carbon atom and the bromine atom • The Br-Br bond breaks by heterolytic fission, with the electron pair going to the bromine • A bromide ion and a carbocation are formed or you could draw this...
Finally, because the positively charged carbocation is unstable, it quickly reacts with the bromide ion to form the organic product 1,2-dibromoethane • In this mechanism, the bonds are broken by heterolytic fission and ions are formed as the intermediates
Curly arrows • You may be asked to define what the curly arrows signify... • Make sure you can draw the reaction of HBr and Br2 with ethene... • So try it! Complete the exam questions on your sheet!
Peer marking • Swap your answers with someone else!
1. (a) curly 1 dipoles shown correctly on the Br–Br and curly arrow from the Br–Br 1 bond towards the Brδ correct intermediate shown 1 curly arrow from the lone pair or the negative charge on the Br - to the C+ 1 (b) (i) Hs are diagonal to each other in the trans/ 1 difference clearly shown in a diagram (ii) (the product is saturated hence) there is no restricted rotation/single 1 bonds allow rotation/because C=C prevents rotation [6]
1. (i) electron/lone pair acceptor 1 (ii) 1 1 1 [4]
But... I’ve got more than one double bond! • Myrcene (C10H16) is a member of the terpene family of chemicals. These are alkenes built from whole numbers of isoprene (C5H6) molecules. • These isoprene molecules can be linked together head to tail forming chains or can be formed into rings.
Tarpenes contain carbon-carbon double bonds. • If reacted with excess hydrogen in the presence of a nickel catalyst, all the double bonds will react and form a saturated molecule
Flow chart • Complete the revision flow chart for the reactions of the alkenes