Understanding Atomic Structure: Essential Questions & Key Discoveries in Chemistry
Explore the fundamental components of atoms and how they shape the universe around us. This guide addresses three critical questions: What are the parts of an atom? How can we differentiate subatomic particles? How does atomic structure impact chemical reactivity? Delve into the rich history of atomic theory, from Democritus's original ideas to Rutherford's groundbreaking experiments. Discover the role of protons, neutrons, and electrons in defining elements and isotopes, along with the significance of valence electrons in chemical bonding.

Understanding Atomic Structure: Essential Questions & Key Discoveries in Chemistry
E N D
Presentation Transcript
The Atom What to look for: Three Essential Questions E. Q. #1 = What are the parts of an atom? E. Q. #2 = In what manner can subatomic particles be differentiated? E. Q. #3 = How does the atomic structure impact chemical change & reactivity?
The Building Block of Matter • 400 BC Democritus (Greek Philosopher) • Atomos = “indivisible” • …like a brick is used to build a building; an atom is used to build all matter…
First Atomic Theory • 1808 John Dalton (English Chemist) • Using experiments formulates the first atomic theory • All elements are composed of indivisible particles called atoms • Atoms of the same elements are exactly the same • Atoms of different elements are different • Two or more elements combine to form compounds
Subatomic Particles • The idea that the atoms was not a solid sphere, but a composition of small particles which came to be known as subatomic particles • Julius Plucker, William Crooks & J J Thomson • mid-1800’s
J J Thomson • 1897 Discovers the electron
Ernest Rutherford (English Physicist) • Gold Foil Experiments • 1908 Discovers that the atom is mostly empty space • Discovers a hard, dense center of the atom called the NUCLEUS
The atom is mostly empty space • Most of the particles passed straight through to gold foil. • First important discovery by Rutherford
The NUCLEUS is Discovered • 1911 Rutherford proves that the particles that do not pass through are hitting a small, dense center of the atom called the nucleus. • Rutherford later goes on to discover the proton
3 Name three major discoveries made by Ernest Rutherford & the Gold Foil Experiment: 1. _______________________________________ 2. _______________________________________ 3. _______________________________________ What are the two results of firing particles at atoms in the foil of the Gold Foil Experiments: 1. _______________________________________ 2. _______________________________________ 2 1 Who was the first to attempt to form a complete atomic theory? 1. _______________________________________
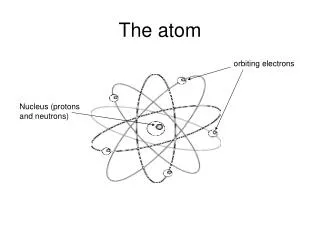
Electron Energy Levels • 1913 Niels Bohr (Danish physicist) discovers the electrons travel around the outside of the atom on different energy levels • The Bohr Model = “solar system” model
The Neutron is Found • 1932 James Chadwick proves the existence of the neutron. • Only 4 years later the neutron is used to split the first atom • Why was the neutron’s existence more difficult to prove? • What evidence hinted of its existence? “the missing mass” E. Q. #1 = What are the parts of an atom?
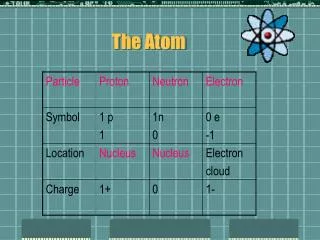
The Subatomic Particles E. Q. #2 = In what manner can subatomic particles be differentiated?
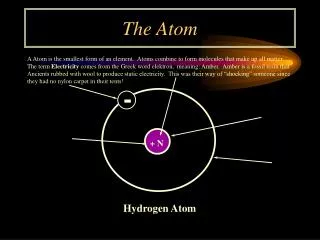
Finding the Number of Subatomic Particles in an Atom • Atomic Number = the number of protons in the nucleus of an atom • Identifies each element • Hydrogen is 1 and it has 1 proton in its nucleus (only hydrogen has 1 proton) • Helium is 2 and it has 2 protons in its nucleus (only helium has 2 protons)
Electrons • IFthe atom is neutral (same number positive charges as negative charges) then the number of electrons will be the same as protons. • ION = a charged atom (unequal number of protons and electrons) • +1 charge = the atom LOST one electron • -1 charge = the atom GAINED one electron • +2 charge = LOST 2 electrons • -2 charge = GAINED 2 electrons and so on
Example of Ions • Sodium will lose one electron when it bonds • Na+1 or Na+ • Sodium’s atomic number is 11, so it has 11 protons. • The +1 charge means Na+1 has 10 electrons
Another Example • Chlorine will gain an electron when it bonds • Cl-1 or Cl- • Chlorine’s atomic number is 17, so it has 17 protons • The -1 charge means Cl-1 has 18 electrons
Mass Number • The two subatomic particles that have mass? • Proton and neutron, both found in the nucleus • Mass Number = # of Protons + # of Neutrons • Mass # - Atomic # = # of neutrons
Practice Problems Do Parts of the Atom & Atomic Structure practice sheet now.
ISOTOPES • Atoms of the same element with a different number of neutrons. • Ex: carbon-12 & carbon-14 • Carbon-12 • 6 protons • 6 neutrons • 6 electrons • Carbon-14 • 6 protons • 8 neutrons • 6 electrons
Isotopes of Hydrogen • Hydrogen-1 (protium) 0 neutrons • Hydrogen-2 (deuterium) 1 neutron • Hydrogen-3 (tritium) 2 neutrons
The ELECTRON CLOUD • The electron cloud is separated by different energy levels. • Electrons with less energy travel on the levels closest to the nucleus. • Energy levels are divided into sub-levels • Sub-levels are divided into orbitals • All of these levels, sub-levels & orbitals can overlap.
VALENCE ELECTRONS • Valence electrons = the outermost electrons • The electrons involved in bonding • Determines the chemical properties of an atom E. Q. #3 = How does the atomic structure impact chemical change & reactivity? Do Practice sheets What Is This Atom Now? 1 & 2 now.
More about Electrons • Maximum # of electrons on each energy level • Level 1 = 2 electrons • Level 2 = 8 electrons • Level 3 = 18 electrons • Level 4 = 32 electrons
“the Stable 8” • Due to the overlapping levels, sub-levels and orbitals the maximum number of outer most electrons is EIGHT. • An atom is becomes stable when it outer-most energy levels is full of electrons • This explains the chemical bonding properties of the atoms • Atoms will do one of three things in order to fill their outer most energy levels. • gain electrons • lose electrons • share electrons
Concepts Coming Up Next • The number of valence electrons an atom has determines the group it becomes to on the PERIODIC TABLE. • Groups or families of elements on the periodic table has similar chemical properties.
Concepts Coming Up Next • Valence electrons determine how atoms will bond with other atoms in order to complete their outer-most energy level. • The Stable 8 • CHEMICAL BONDING • Ionic bonding (gaining/losing of electrons) • Covalent bonding (sharing of electrons)