Cell Respiration
240 likes | 403 Views
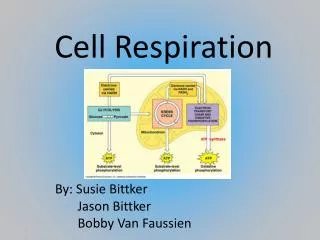
Cell Respiration. Topics 3.7 & 8.1. Definition of cell respiration. Cell respiration is the controlled release of energy from organic compounds in cells to form ATP. It takes place in ALL cells – prokaryotic and eukaryotic. It can be aerobic (involving oxygen) or anaerobic (no oxygen).

Cell Respiration
E N D
Presentation Transcript
Cell Respiration Topics 3.7 & 8.1
Definition of cell respiration • Cell respiration is the controlled release of energy from organic compounds in cells to form ATP. • It takes place in ALL cells – prokaryotic and eukaryotic. • It can be aerobic (involving oxygen) or anaerobic (no oxygen).
Anaerobic Respiration • The process of respiration takes places in several steps (metabolic pathway). • Anaerobic respiration occurs in the CYTOPLASM. • Glucose is broken down into a simpler substance called PYRUVATE. (pyruvic acid) • A small amount of ATP is produced in this reaction.
Anaerobic respiration contd. • If no O2 is available, the pyruvate is converted into waste products that are later removed from the cell. • In humans the waste product is LACTATE (lactic acid). • In yeast the waste products are ETHANOL and CARBON DIOXIDE. • No further ATP is made.
In humans In yeast Glucose Glucose Pyruvate Pyruvate Lactate Ethanol + CO2 Small yield of ATP Small yield of ATP Anaerobic respiration contd. GLYCOLYSIS GLYCOLYSIS
Aerobic respiration • If O2 is available, the pyruvate enters the mitochondria where it is broken down into CO2 and water. • A large amount of ATP is produced in these reactions. CO2 + H2O Pyruvate Large yield of ATP
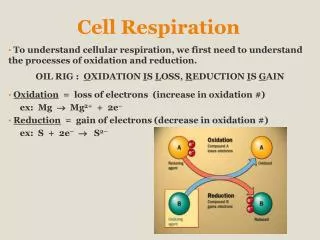
Oxidation & Reduction in Cell Respiration • Cell respiration involves several redox reactions. • Hydrogen carriers accept hydrogen atoms removed from substrates. • NAD+ + 2H NADH + H+
Glycolysis • First step in both aerobic and anaerobic respiration. • Occurs in cytoplasm. • Four steps:- • Phosphorylation – 2 phosphates are added to glucose to form hexose biphosphate. 2ATPs provide the phosphates and the resulting molecule now has a higher energy level.
Glycolysis contd. • Lysis – hexose biphosphate splits into 2 molecules of triose phosphate. • Oxidation – 2 hydrogen atoms removed from each triose phosphate and collected by NAD+. • ATP formation – Pyruvate is formed by removal of 2 phosphates that are joined to ADP to make ATP.
Glucose Summary One glucose is converted into 2 pyruvates 2 NAD+ are converted into 2 NADH + H+ 2 ATP molecules used per glucose but 4 are produced giving a net yield of 2 ATP. Glycolysis diagram 2 ATP PHOSPHORYLATION 2 ADP Hexose biphosphate LYSIS 2 triose phosphates 2 NAD+ OXIDATION 2 NADH + H+ 4 ADP ATP FORMATION 4 ATP 2 pyruvates
Aerobic respiration – link reaction • Pyruvate from glycolysis is absorbed by the mitochondrion. • The pyruvate is both OXIDIZED and DECARBOXYLATED. • Enzymes in the matrix of the mitochondrion are responsible for each process. • The hydrogen that is removed is accepted by NAD+ to form NADH + H+. • The decarboxylated pyruvate is a 2-carbon compound (acetyl group) that reacts with coenzyme A.
Summary of the link reaction OXIDATION Pyruvate acetyl CoA NADH + H+ NAD+ Coenzyme A CO2 DECARBOXYLATION
Aerobic respiration – Kreb’s Cycle • An acetyl group (CH3CO) is transferred from acetyl CoA to a 4-carbon compound (oxaloacetate). • This results in the production of a 6-carbon compound (citrate). • Citrate is converted back into oxaloacetate by a series of reactions involving DECARBOXYLATION, OXIDATION and PHOSPHORYLATION.
Kreb’s cycle contd. • CO2 is removed in 2 of the reactions as a waste product and excreted together with CO2 from the link reaction. • Hydrogen is removed in 4 of the reactions. The hydrogens are picked up by carriers (NAD+ and FAD). • These oxidations release energy which is stored by the carriers when they accept hydrogen. • This energy is later released by the electron transport chain and used to make ATP. • ATP is produced directly in one of the reactions.
NAD+ NAD+ NADH + H+ NADH + H+ CO2 CO2 Summary of the Kreb’s cycle acetyl CoA CoA oxaloacetate (C4) citrate (C6) NADH + H+ NAD+ FADH2 C5 FAD C4 ATP ADP
The electron transport chain • The electron transport chain (ETC) is a series of electron carriers, located in the inner membrane of the mitochondrion. • NADH supplies 2 electrons (e-) to the first carrier. • These e- pass along the chain, giving up energy at each stage.
The electron transport chain contd. • At 3 points along the chain enough energy is given up for ATP to be made by the enzyme ATP synthetase. • This process is called OXIDATIVE PHOSPHORYLATION. • FADH2 also feeds e- into the ETC, but at a later stage than NADH, resulting in 2 ATP molecules instead of 3.
ADP ATP ADP ATP ADP ATP The final recipient of these e- is oxygen. Oxygen is reduced to form water. This is the only stage of cellular respiration that uses oxygen. Summary of ETC NADH NAD FADH FAD Reduced cytochromes Oxidized cytochromes Reduced cytochrome oxidase Oxidized cytochrome oxidase H2O O2
Chemiosmosis • The energy released as e- pass along the ETC is used to pump protons (H+ ions) from the matrix into the intermembrane space. • Due to the small volume of this space, it quickly becomes concentrated with protons. • This creates 2 areas with different proton concentrations – LOW in matrix, HIGH in intermembrane space.
Chemiosmosis contd. • Protons move down the concentration gradient by passing through channels provided by the enzyme ATP synthase, found in the inner membrane. • When the ATP synthase enzyme is activated, ADP is phosphorylated into ATP. • The coupling of ATP synthesis to electron transport is called CHEMIOSMOSIS.
Relationship between structure & function of mitochondria • Cristae – foldings of the inner membrane that increase the surface area for the electron transport chain and oxidative phosphorylation. • Fluid matrix – contains enzymes for link reaction and Kreb’s cycle. • Intermembrane space – the space between inner and outer membranes is small to allow for accumulation of protons for chemiosmosis.
The End • Now all you have to do is….. • ……LEARN IT