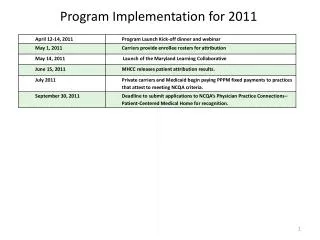
Program implementation
Program implementation. Pascale Wortley, MD, MPH January 22, 2010 Program Managers Meeting. H1N1 Response: overall structure. Deputy Incident Manager. Incident Manager. Chief of Staff. Chief Health Officer. Epi/surveillance Task Force. Vaccine Task Force. Medical

Program implementation
E N D
Presentation Transcript
Program implementation Pascale Wortley, MD, MPH January 22, 2010 Program Managers Meeting
H1N1 Response: overall structure Deputy Incident Manager Incident Manager Chief of Staff Chief Health Officer Epi/surveillance Task Force Vaccine Task Force Medical Countermeasures Task Force Community Mitigation Task Force State Coordination Task Force
H1N1 Vaccine Task Force Task ForceSenior Deputy for ScienceCynthia Whitney Task Force Director Jay Butler Task ForceChief of StaffDave Brownell H1N1 Vaccine Steering Committee Vaccine Task Force DeputyTom Shimabukuro Vaccine Data Management TeamDavid Walker Vaccine Task Force Operations Analyst Eric Marble Doses Administered TeamJeanne Tropper Warren Williams Vaccine Coverage & Monitoring team Jim Singleton Gary Euler Vaccine Implementation TeamPascale Wortley Vaccine DistributionTeamJeanne Santoli Vaccine Communication& Education TeamKris Sheedy VaccineSafety TeamClaudia VellozziFrank DeStefano Vaccine Effectiveness Team David Shay Vaccine EOC DeskDuane KilgusVicki Evans
Status Update • Doses ordered to date (Sept 31-January 15): 120 million doses • VFC program annual ordering: 80 million doses • Almost 120,000 provider agreements signed • Almost 70,000 unique ship-to sites
Cumulative Number of Provider Agreements, H1N1 Vaccine Program, Sept 2009-Jan 2010 Pre-launch
Cumulative Number of Ship-to Sites, H1N1 Vaccine Program, Oct 2009-Jan 2010
As of 15 Jan 2009 Pro-Rata Allocation: 139,334,000 Advance Allocation: 5,691,500 Retail Allocation: 5,521,800
Number of Doses Shipped From Each Depot (7-Day Moving Averages) CA Depot: AZ, CA, CO, HI, NV, OR, UT, WA, Pacific Territories OH Depot: IN, MI, MN, NE, NJ, NY, NYC, OH, PA, Philly, WI
Number of Doses Shipped From Each Depot (7-Day Moving Averages) GA Depot: AL, CT, DE, FL, GA, KY, ME, MD, MA, NH, NC, RI, SC, TN, VA TN Depot: AK, AR, DC, ID, IL, Chicago, IA, KS, LA, MS, MO, MT, NM, ND, OK, PR, SD, TX, VT, VI, WV, WY, Federal Employees
Reported intention to be vaccinated Note: Among the general population, 43% of those who intend to get vaccinated for H1N1 have already done so.
H1N1 Influenza Vaccination Coverage by ACIP Target Groups, NHFS Dec 27, 2009-Jan 02, 2010 *Estimate may not be reliable, (CI half-width/estimate)≥0.6 or (CI half-width)≥10. **Estimates with an unweighted denominator of <30 observations are not reported and are coded as NA.
H1N1 Influenza Vaccination Coverage - childrenSource: National H1N1 Flu survey • Month of Dec 2009 - picture of mid Dec Age • 6-23 months 33.4±6.4 • 2-4 years 30.0±5.5 • 5-18 years 24.2±3.2 • 5-9 years 32.6±5.8 • 10-12 years 21.8±5.6 • 13-18 years 18.7±4.6
Note: The proportion of people in non-initial target groups receiving H1N1 vaccine has increased as states have relaxed prioritization.
Note: In some weeks, black non-Hispanics appear to have somewhat lower coverage rates than other groups.
Cumulative H1N1 influenza vaccination coverageamong children aged 6 months-17 years, by statebased on combined estimates from the December 2009 BRFSS and NHFS interviews Combined national estimate=27.6% Significantly >30%: Indiana, Massachusetts, Connecticut, South Dakota, Maine, Rhode Island. Significantly >20%: Washington, New York, Ohio, Virginia, Nebraska, Kansas, Iowa, Delaware, Hawaii, New Hampshire, Pennsylvania, Minnesota, Illinois, West Virginia, South Carolina, Vermont, Arkansas. Significantly <20%: Texas.
Cumulative H1N1 influenza vaccination coverageamong persons 6 months and older, by statebased on combined estimates from the December 2009 BRFSS and NHFS interviews National estimate: 13.6% (12.5-13.7) Range: 6.2-23.1%
Note: In December, more people reported receiving H1N1 vaccine in non-medical locations, particularly schools and worksites, than in October and November. Totals do not equal 100% due to “don’t know”
Weekly uptake of seasonal and H1N1 influenza vaccines through January 02, 2010, SDI Note: An uptick in billing for seasonal and H1N1 influenza vaccinations in physician offices occurred after the Thanksgiving holiday week, but decreased again during the Christmas holidays.
H1N1 Influenza Vaccine Uptake in Physician Offices, through January 2, 2010, SDI Note: Observed dips in billing for H1N1 vaccinations represent Thanksgiving and Christmas holiday weeks.
Next steps • Completing the campaign, determining how to scale back efforts • Evaluation: what are the most important things to learn • for seasonal vaccination • for future responses