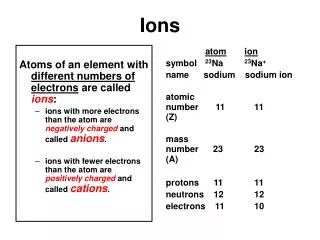
Ions
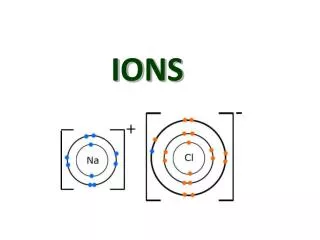
Ions. Ions : A charged particle formed when a neutral atom or group of atoms gain or lose one or more electrons. Example Na Na + + e - F + e - F -. Cations. Mg Mg 2+ + 2e - Al Al 3+ + 3e - Cation: A positively-charged ion. One or more electrons are lost from a neutral atom

Ions
E N D
Presentation Transcript
Ions Ions: A charged particle formed when a neutral atom or group of atoms gain or lose one or more electrons. Example Na Na+ + e- F + e- F-
Cations Mg Mg2+ + 2e- Al Al3+ + 3e- Cation: A positively-charged ion. One or more electrons are lost from a neutral atom oxidation
Anions I + e- I- O + 2e- O2- S + 2e- S2- Anion: A negatively-charged ion. Electrons are gained by a neutral atom reduction
Ions • Ion charges can be predicted from the Periodic Table • Main Group metal (IA-IVA) ion-charges correspond to group number • Sodium (Na) in IA +1 • Exceptions • Tl, Sn, Pb, Sb, and Bi • We’ll talk about non-main group metal ion-charges later • Main Group non-metal (IIIA-VIIIA) ion-charges correspond to (group# - 8) • Fluorine (F) in VIIA (7-8) = -1 • Boron is exception -3 not -5 • All this has to do with electron configuration • More anon • See website as well: http://web.clark.edu/aaliabadi/CHEM131ions%20to%20memorize.htm
Compounds That Contain Ions • Require metal and non-metal • Form ionic bonds • Called an ionic compound Characteristic Properties 1. Very high melting points 2. Conduct an electric current when melted or when dissolved in water
Ionic compound • The number of cations and anions must have a net charge of zero.
Compounds that Contain Ions Writing Formulas for Ionic Compounds Give the formulas for the compounds that contain the following pairs of ions: (a) K and I (b) Mg and N (c) Al and O
Types of Ionic Compounds • Type I Compounds: The metal present forms only one type of cation. • Examples: the main group metals • Type II Compounds: The metal present can form two or more cations that have different charges or oxidation states • Oxidation state = ionic charge, if any, on species • Examples include Cr2+, Cr3+, Cu+, Cu2+, etc.
Naming Ionic Compounds 1. The cation is always named first and the anion second. 2. The cation takes its name from the name of the element. 3. The anion is named by taking the first part of the element name and adding –ide.
Name the following Type I compounds NaCl RaBr2 Rb2O AlI3 K3N Cs4Si Give the chemical formula for the following Type I compounds Strontium phosphide Calcium fluoride Beryllium carbide Lithium hydride Barium sulfide Magnesium telluride Naming Ionic Compounds
Naming Ionic Compounds • Type II compounds need to be identified by a Roman numeral (I), (IV), etc. • Represents oxidation state of cation • Not how many cations are present in compound! • Example: NaCl sodium (I) chloride is INCORRECT • Example: SnCl4 tin (IV) chloride is correct
Naming Ionic Compounds Type II Ionic Compounds FeCl2 and FeCl3 PbO and PbO2 MnS and Mn2S7
A Mixed Bag • PbBr2 and PbBr4 • Aluminum arsenide • FeS and Fe2S3 • Thallium (III) boride • Mercury (II) carbide • Na2S • CoCl3 • Cerium (IV) phosphide • ScF3 • Gold (I) selenide • Vanadium (V) telluride
Naming Compounds that Contain Polyatomic Ions Polyatomic Ion: An ion that contains more than one atom. They are charged entities composed of several atoms bound together. • Consult my website for the list that must be memorized: • http://web.clark.edu/aaliabadi/CHEM131ions%20to%20memorize.htm
Trends • Sulfide, sulfite, sulfate • Nitride, nitrite, nitrate • Phosphide, phosphite, phosphate • Chloride, hypochlorite, chlorite, chlorate, perchlorate • Parenthesis required if more than one polyatomic ion present • Ca(IO3)2 is correct • Ca(I)2 is INCORRECT
Naming Compounds that Contain Polyatomic Ions Name or provide the chemical formula for each of the following compounds: (a) Ca(OH)2 (e) Co(ClO4)2 (b) Sodium phosphate (f) platinum (IV) bicarbonate (c) KMnO4 (g) Cu(NO2)2 (d) Ammonium dichromate (h) nickel (III) oxalate
Naming Compounds that Contain Polyatomic Ions • Name or provide each of the following compounds: (a) calcium carbonate (e) MoO (b) BaSO4 (f) Iridium (VII) acetate (c) CsClO4 (g) ZnHPO4 (d) Zirconium bisulfite (h) lithium cyanide
Naming Acids Acids: A substance that yields hydrogen ions (protons, H+) when dissolved in water. • HCl(aq) H+(aq) + Cl-(aq) • H3PO4(aq) 3H+(aq) + PO43-(aq)
Rules for naming acids • If the formula does not contain oxygen the prefix of the acid is hydro and the suffix –ic is attached to the root name for the element. • Ex: HCl = hydrochloric acid, H2S = hydrosulfuric acid • When the anion contains oxygen, the acid name is formed from the anion name. The suffix –ic or –ous is added. • When the anion ends in –ate, the suffix –ic is used. • H2CO3 = carbonic acid • When the anion ends in –ite, the suffix –ous is used. • H2SO3 = sulfurous acid
More AnionAcid ClO4- ______________ HClO4 _______________ ClO3- ______________ HClO3 _______________ ClO2- ______________ HClO2 _______________ ClO- _______________ HClO ________________
Naming Compounds that Contain Only Nonmetals: Type III • Type III compounds contain only nonmetals. • Form covalent bonds • share electrons Rules for Naming Type III Binary Compounds 1. The first element in the formula is named first, and the full element name is used. 2. The second element is named as though it were an anion. 3. Prefixes are used to denote the numbers of atoms present. 4. The prefix mono- is never used for naming the first element. 5. Drop the “a” when it is followed by an “o” Tetraoxide should be tetroxide
Naming Compounds that Contain Only Nonmetals: Type III Prefixes Used to Indicate Numbers in Chemical Names PrefixNumber Indicated mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10
Practice • CCl4 • Silicon dioxide • NO2 • Sulfur trioxide • P2O5 • Iodine pentafluoride • Dinitrogen tetroxide • SeI2 • Xenon hexafluoride
The Name Game • Extra credit opportunity • Mix-and-match
Remember the mole? • Mole = amt that contains as many “things” as there are atoms of 12 g of C-12 • 1 mole = 6.022 x 1023 particles • Molar mass (MM) = mass in grams per 1 mole of particle (g/mol)
Molecular Mass • Summation of molar masses from Periodic Table based on molecular formula • NaCl • I2 • V2O5
Molar Mass Example Calculate the mass of 30.0 moles of polyvinyl chloride (PVC), C2H3Cl.
Molar Mass Example A sample of Na2SO4.with a mass of 300.0 grams represents what number of moles of Na2SO4? Example Calculate the number of grams of caffeine, C8H10N4O2, in 8.13 x 1023 molecules.
Percent Compositionof Compounds Example Compute the mass percent of each element in sodium sulfide, Na2S. Example Compute the mass percent of each element in nitric acid, HNO3(aq).
Empirical Formulas Empirical Formula: or the simplest formula; the smallest whole-number ratio of the atoms present. Molecular Formula: the actual formula of a compound. It gives the composition of the molecules that are present. Empirical FormulaMolecular Formula CH C6H6 CH2O C6H12O6 H2O H2O
Calculation of Empirical Formulas Steps for Determining the Empirical Formula of a Compound 1. Obtain the mass of each element present (in grams). 2. Determine the number of moles of each type of atom present. 3. Divide the number of moles of each element by the smallest number of moles to convert the smallest number to 1. If all of the numbers are integers (whole numbers), these are the subscripts in the empirical formula. If one or more of these numbers are not integers, go to step 4. 4. Multiply the numbers derived in step 3 by the smallest integer that will convert all of them to whole numbers. This set of whole numbers represents the subscripts in the empirical formula.
Calculation of Empirical Formulas Experiment: Suppose we weigh out 6.50 grams of Cr. We decide to heat this Cr in the air so that the Cr can react with O to form CrxOy . After the sample cools, we weigh it again and find its mass to be 9.50 grams. How do we find the mass of oxygen? What is the empirical formula of this compound? Let’s work on this together.
Calculation of Empirical Formulas Example Nylon-6 consists of 63.68% C, 12.38% N, 9.80% H, and 14.14%O. Calculate the empirical formula for Nylon-6.
Calculation of Molecular Formulas We need to know the empirical formula and molar mass of the molecular compound. Molecular Formula = (empirical formula)n where n is a small whole number. Molecular Formula = n x Empirical Formula Molar Mass = n x Formula Weight n = Molar Mass/Formula Weight
Calculation ofMolecular Formulas Example A compound shows the following percentage compostion: 71.65% Cl 24.27% C 4.07% H The molar mass is known to be 98.96 g/mol. Determine the empirical formula and the molecular formula for this compound.
Calculation ofMolecular Formulas Example Vitamin C consists of 40.92% C, 4.58% H, and 54.50% O on a mass basis, and has a molar mass of 176.12 g/mol. Determine the molecular formula of the compound.