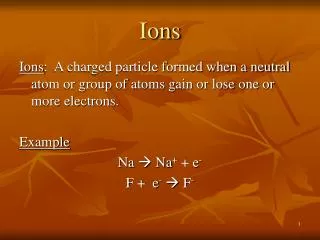Ions
Ions. Go electrons!. Ions. Atoms like to have a full valence shell. In order to do this, often times atoms need to gain or lose electrons. When an atom gains or loses an electron, the atom becomes an ION!. Transfer of Electrons.

Ions
E N D
Presentation Transcript
Ions Go electrons!
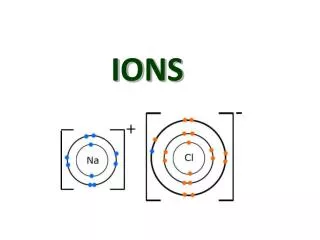
Ions • Atoms like to have a full valence shell. • In order to do this, often times atoms need to gain or lose electrons. • When an atom gains or loses an electron, the atom becomes an ION!
Transfer of Electrons An electron doesn’t ever just fly off into space, when one atom loses an electron, another atom must gain it!
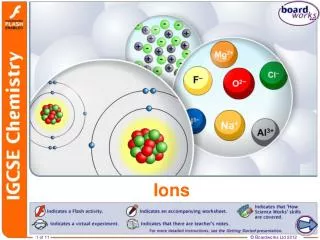
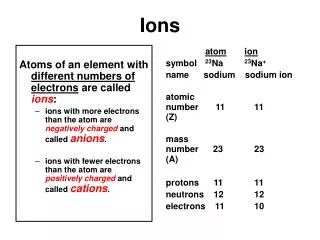
More about ions! • What charge do electrons have? • …..yep….that’s right! Negative! • So, if an atom loses one or more electrons, what kind of charge will it have? • ….yep! Positive charge. Ions with a positive charge have more protons than electrons and are called CATIONS!
And more about ions • So, then what if an atom gains some electrons? What charge will it have? • YES! A negative charge because there are more electrons than there are protons. A negative ion is called an ANION!
Charges on ions… • Let’s look at a few atoms and figure out what kind of charge they would have. • First look up Fluorine…. • How many electrons are in its outer (valence) shell? • Would it be easier for it to gain or lose electrons to have a full outer shell? • Because it already has 7 electrons in its outer shell, it is going to be much easier to gain one electron, than to lose 7 electrons. • Is it going to become a positive or negative ion?
Practice Time • Figure out what the charges would be on the following atoms: • Magnesium • Bromine • Potassium • Arsenic • Iodine