PERIODIC TRENDS
PERIODIC TRENDS. Unit 5, Periodic Trends and Electron Configuration. s, p, d, f Blocks. Remember in the lab we did with electron configuration, there was a pattern on the periodic table.

PERIODIC TRENDS
E N D
Presentation Transcript
PERIODIC TRENDS Unit 5, Periodic Trends and Electron Configuration
s, p, d, f Blocks Remember in the lab we did with electron configuration, there was a pattern on the periodic table. This pattern is the basis for modern chemistry. While protons define an atom, electrons define the atom’s reactivity.
Why does this matter? Valence electrons are those electrons found in the highest energy level in an atom. All elements in a group have the same number of valence electrons in their highest energy level.
Problems Given each of the following valence electron configurations, determine which block of the periodic table the element is in: • s2p4 b. s2d1 c. s1 d. s2p1
Describe how each are related: Group number and number of valence elections: Group number in Roman numerals (with A) is the same as the number of valence electrons. Principal energy level of valence electrons and period number: Period number is the energy level in which you find valence electrons for elements in that period.
Electron Configurations • Don’t have to write out the entire electron configuration. • There is a short-cut: • Keeps focus on valence electrons • An atom’s inner electrons are represented by the symbol for the nearest noble gas with a lower atomic number. K: [Ar]4s1
Electron Configurations For the element Phosphorus -- 15 electrons 3s23p3 1s22s22p63s23p3 P: [Ne] Must be a Noble gas (One just before Element)
Electron Configurations Let’s do a couple more: Ba: [Xe] 6s2 Hg: [Xe] 6s2 5d10 4f14 V: [Ar] 4s2 3d3
PNICOGENS CHALCOGENS
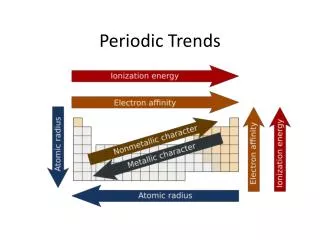
Periodic Properties • An element’s properties can go hand in hand with electron arrangement • We can use an element’s location on the PT to predict many properties. • Atomic radius • Ionic Size • Ionization energy • Electronegativity
99 pm 198 pm Atomic Radius • The radius of an atom is defined by the edge of its last energy level. • However, this boundary is fuzzy • An atom’s radius is the measured distance between the nuclei of 2 identical atoms chemically bonded together - divided by 2.
Atomic Radius • As we examine atomic radius, we see a gradual decrease in atomic size from left to right across the PT. • As e- are added in the same energy level, they are pulled closer to the highly positive nucleus.
Atomic Radius • Atomic radius increases going down a family. • The change in atomic radii going down is due to e-shielding • As we move down a group on the PT, we add an energy level of e-‘spushing the valence e’s farther away from the nucleus.
Periodic Properties • How does the size of an atom change when electrons are added or removed? As an atom loses 1 or more electrons (becomes positive), it loses an energy layer therefore, its radius decreases.
Ion Radius • How does the size of an atom change when electrons are added or removed? As an atom gains 1 or more electrons (negative), it fills its valence energy level, therefore, its radius increases.
Loses 1 electron 4s 4s [Ar] K: [Ar] +1 K Wants a full set of e- Positive Ion Radius Properties • Elements in a group tend to form ions of the same charge. • Modeled by electron configurations.
Gains 2 electrons 2s2 2p4 Wants a complete set -2 O Negative Ion Radius O: [He] [He]
18 1 2 13 14 15 16 17 3 4 5 6 7 8 9 10 11 12 +1 tend to have +2 -2 -1 +3 -3 +/- 4 more than one option + 3 + 3 or + 4 Periodic Trend of Ionic Charges
Ion Radius Trends Ion radius increases going down a group due to added energy levels. Ion radius across a period depends on the charge of the ion; positive ions are smaller than their neutral atoms (due to missing energy level) and negative ions are larger than their neutral atoms (due to increased repulsion of the added electrons.
Tend to lose electrons to become positive Tend to gain electrons to become negative
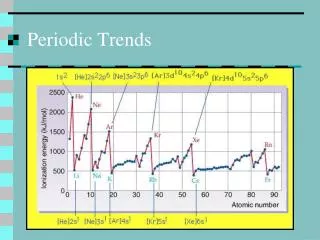
Ionization Energy Properties The energy needed to remove an atom’s electron. • If the e-s are held strongly the atom will have a high ionization energy • The first electron is always the easiest to remove. • The more valence electrons, the more difficult to remove one. • Noble gas configurations are exceptionally high.
Ionization Energy • IE decreases down a group due to the increasing number of energy levels shielding the valence electrons. • IE increases left to right across a period because as an atom gets closer to an octet, the attraction for valence electrons increases • There is generally a large jump in energy necessary to remove additional electrons from the atom.
The amount of energy required to remove a 2p e– (an e- in a full sublevel) from a Na ion is almost 10 times greater than that required to remove the sole 3s e-
Electronegativity Electronegativity reflects the ability of an atom to attract electrons in a chemical bond. --Decreases going down a group due to shielding from added energy levels. • Increases left to right across a period due to increased strength of attraction between nucleus and valence electrons. Electronegativity correlates to an atom’s ionization energy.
Electronegativity Electronegativity
Electron Dots Draw the symbols of atoms with dots to represent the valence-shell electrons for period 3. 1A 2A 3A 4A 5A 6A 7A 8A Na MgAlSiP S ::Cl :Ar:
Bonding • Atoms are generally found in nature in combination held together by chemical bonds. • A chemical bond is a mutual electrical attraction between the nuclei and outer electrons of different atoms that binds the atoms together so that they behave as one unit. • There are two types of chemical bonds: ionic and covalent.
Introduction to Ionic Bonding • Two atoms in an ionic bond transfer electrons from one element to another. • Generally, electrons move from a metal to a nonmetal. • The electron is transferredfrom the atom with the low electronegativity to the atom with the high electronegativity.
Introduction to Covalent Bonding • Two atoms involved in the covalent bond share electrons in order to achieve the arrangement of a noble gas. • Generally between two nonmetals. • Because electronegativities are similar so neither wants to give up electrons.
Rule of Thumb metal w/nonmetal = usually ionic nonmetal w/nonmetal = usually covalent
Metallic Bond • The electron sea model proposes that all the atoms in a metallic solid contribute their valence electrons to form a “sea” of electrons. • A metallic bond is the attraction of a metallic ion for the “sea” of delocalized electrons.
Metals Form Alloys • Metals do not combine chemically with metals. They form alloyswhich are solutions of a metal in a metal. • Delocalized electrons are free to move heat or electricity easily through metals. • Number of delocalized electrons and strength of metallic bond determine melting point.
Types of Alloys There are two kinds of alloys. Substitutional alloys are made of elements of similar size (sterling silver, brass, pewter). Interstitial alloys are made of elements with different size atoms so that little ones fit between larger ones (carbon steel).
(stop) Dogs Teaching Chemistry Video Bonding Video