Atomic Structure
Atomic Structure. Characteristic spectrum. Continuous spectrum. X-ray Spectrum. Atomic Structure. Atoms have electrons in energy levels of increasing energy outer electrons are removed more easily than the inner electrons consider an electron of kinetic energy K passing close to an atom

Atomic Structure
E N D
Presentation Transcript
Characteristic spectrum Continuous spectrum X-ray Spectrum
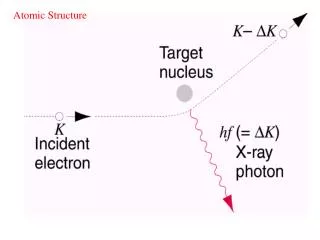
Atomic Structure • Atoms have electrons in energy levels of increasing energy • outer electrons are removed more easily than the inner electrons • consider an electron of kinetic energy K passing close to an atom • a “collision” in which the electron loses kinetic energy which appears as the energy hf of a photon which radiates away from the atom • x-rays are emitted ( bremsstrahlung) • there is a minimum wavelength. Why?
X-ray spectrum • If electron loses all its energy, eVaccel= hfmax = hc/min • min is independent of the material and depends only on KE of electrons • note that if h=0, then min =hc/eVaccel would be zero! • the peaks at larger depend on the material • arise when the incident electron knocks out an inner electron • this leaves a hole in an inner shell which is filled by an outer electron with the emission of an x-ray photon Note K K lines
M shell => n=3 L shell => n=2 K shell => n=1
Moseley Plot • Moseley (1913) measured characteristic x-rays of as many elements as he could find at the time • he found that he could order the elements by atomic number Z rather than by atomic weight (i.e. increasing number of electrons) • for the Khe plotted the square root of frequency vs position in periodic table and found a straight line • data could be fit to
Bohr Theory • Characteristic x-ray spectrum identifies elements • depends on Z which determines the chemical properties • K-shell electrons are close to nucleus • visible spectrum involves transitions of outer electrons • Bohr theory works for hydrogen but not multi-electron atoms • however it works well for the Moseley plot • consider an L-electron (n=2 level) about to make a transition to the K-shell which now only has one electron left • L electron “sees” a net charge of Ze + (-e) = (Z-1)e • more precise calculations find (Z-b)e where b~1 • Bohr theory for a transition E between n=2 and n=1 levels
Bohr Theory • Replace Z by (Z-b) ~ (Z-1) • Agrees fairly well with the experimental data for K-lines • does not work well for L-lines • need quantum mechanical treatment • does not work well at higher values of Z
Properties of Light • Sunlight is composed of many wavelengths Continuous visible spectrum Line spectra from H, He, Ba, Hg
Photon-Atom Interactions Energy of photon too small f `=f Scattered photon has f ` < f hf just matches E Atom excited to higher level and makes several transitions Electron escapes and photon absorbed
Photon-Atom Interactions Much higher energy and a photon is emitted Atom in excited state and hf matches E Outgoing photon is in phase with incident photon and in same direction => more photons! Light from different atoms is coherent
Incoherent and not monochromatic Incoherent and monochromatic Coherent and monochromatic
Lasers • Light amplification by stimulated emission of radiation • produces a beam of coherent photons by stimulated emission laser
Ruby Laser Normally all atoms are in the ground state E1 For the laser to work, we need more atoms in an excited state --> called population inversion Optical pumping is used to excite electrons to higher levels which then relax to the state E2
Particle picture Wave picture