7.6 Entropy Change in Irreversible Processes
190 likes | 884 Views
7.6 Entropy Change in Irreversible Processes.

7.6 Entropy Change in Irreversible Processes
E N D
Presentation Transcript
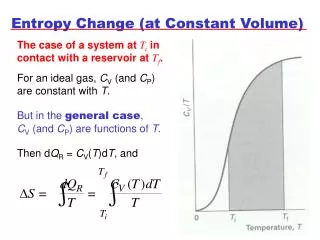
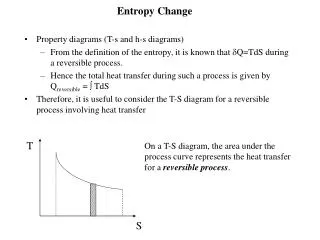
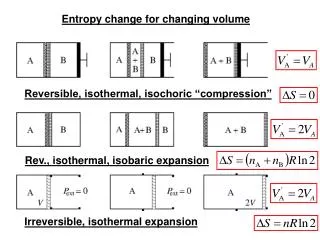
7.6 Entropy Change in Irreversible Processes • It is not possible to calculate the entropy change ΔS = SB - SA for an irreversible process between A and B, by integrating dq / T, the ratio of the heat increment over the temperature, along the actual irreversible path A-B characterizing the process. • However, since the entropy is a state function, the entropy change ΔS does not depend on the path chosen.The calculation of an irreversible process can be carried out via transferring the process into many reversible ones: • Three examples will be discussed here: (1) heat exchange between two metal blocks with different temperatures; (2) Water cooling from 90 to a room temperature; (2) A falling object.
Thermodynamics Potential Chapter 8
8.1 Introduction • Thermodynamic potentials: Helmholtz function F and the Gibbs function G. • The enthalpy, Helmholtz function and Gibbs functions are all related to the internal energy and can be derived with a procedure known as Legendre differential transformation. • The combined first and second laws read dU = Tds – PdV where T and S, and -P and V are said to be canonically conjugate pairs. • By assuming U = U(S,V), one has
8.2 The Legendre Transformation • Consider a function Z = Z(x, y), the differential equation is dZ = Xdx + Ydy where X and x, Y and y are by definition canonically conjugate pairs. • We wish to replace (x, y) by (X, Y) as independent variables. This can be achieved via transforming the function Z(x,y) into a function M(X,Y). • Assume M(X,Y) = Z(x,y) – xX – yY Then dM = dZ – Xdx – xdX –Ydy – ydY dM = -xdX - ydY
8.5 The Helmholtz Function • The change in internal energy is the heat flow in an isochoric reversible process. • The change in enthalpy H is the heat flow in an isobaric reversible process. • The change in the Helmholtz function in an isothermal reversible process is the work done on or by the system. • The decrease in F equals the maximum energy that can be made available for work.
8.6 The Gibbs Function • Based on the second law of thermodynamics dQ ≤ T∆S with dQ = ∆U + P ∆V • Combine the above expressions ∆U + P ∆V ≤ T∆S ∆U + P ∆V - T∆S ≤ 0 • Since G = U + PV –TS (∆G)T,P≤ 0 at constant T and P or G f ≤ Gi • Gibbs function decreases in a process until a minimum is reach, i.e. equilibrium point. • Note that T and P need not to be constant throughout the process, they only need to have the same initial and final values.