THE EARTH’S ATMOSPHERE
850 likes | 1.16k Views
THE EARTH’S ATMOSPHERE. The atmosphere of Earth consists of several regions or layers having different properties (temperature, pressure, and composition) which vary with altitude in different ways.

THE EARTH’S ATMOSPHERE
E N D
Presentation Transcript
THE EARTH’S ATMOSPHERE • The atmosphere of Earth consists of several regions or layers having different properties (temperature, pressure, and composition) which vary with altitude in different ways. • The lowest portion of the atmosphere, the troposphere, contains most of the atmospheric mass, is the part of most direct importance to life on Earth, and is the subject of the field of atmospheric science known as meteorology. • The next highest layer of the atmosphere, the stratosphere, is important because it contains the ozone layer which shields life on Earth from harmful solar ultraviolet radiation. • The higher layers of the atmosphere, the mesosphere and the thermosphere, differ significantly in composition from the lower regions, and also contain a significant proportion of ionized gas which constitutes the ionosphere. • The ionosphere is of practical importance because it facilitates long-distance radio communications. • The field of atmospheric science concerning the mesosphere and upper atmosphere is aeronomy.
EARLY HISTORY OF EARTH’S ATMOSPHERE • Earth’s original atmosphere was quite different from the present atmosphere; it probably consisted mostly of nitrogen (N2) and carbon dioxide (CO2) with much smaller amounts of reduced gases such as carbon monoxide (CO), methane (CH4), ammonia (NH3) and hydrogen sulfide (H2S). • It is also possible that very early in Earth’s history, significant amounts of hydrogen (H2), the most abundant part of the material from which the solar system was formed, could have been a major part of the atmosphere. • Water (H2O), currently mostly liquid in Earth’s oceans, has about 300 times the mass of Earth’s current atmosphere. If temperatures on the early Earth were sufficiently high, H2O would have been the primary constituent of the atmosphere (about 300 bars), followed by CO2 (70 to 90 bars). • Most of the original CO2 is now locked up in carbonate rocks, such as limestone (calcium carbonate, CaCO3), as a result of water erosion (as carbonic acid, H2CO3) of the original silicate rocks: (Ca, Mg)SiO3 + H2CO3 (Ca, Mg)CO3 + H2O + SiO2 • The “noble” gases, such as helium, neon, and argon in the atmosphere, are primarily produced by the decay of radioactive elements.
EARLY HISTORY OF EARTH’S ATMOSPHERE • Both fossil and mineral evidence indicate that oxygen (O2) was only a minor constituent of the atmosphere for the first half of Earth’s existence (the Archean eon), and did not reach near-current levels until the Phanerozoic eon (beginning 600 million years ago). • The primary source of Earth’s atmospheric oxygen is green plant photosynthesis, in which carbon dioxide and water are combined to produce carbohydrates (and molecular oxygen is a “waste product”). • In the first half of Earth’s early history, only photosynthetic bacteria (cyanobacteria, or “blue-green algae”), which did not require oxygen themselves (also known as anaerobic bacteria), were available for photosynthesis.
INVENTORY OF CARBON NEAR EARTH’S SURFACE(Relative Units) Biosphere 2 Atmosphere (as CO2) 70 Oceans (as dissolved CO2) 4000 Fossil Fuels 800 Shales 800,000 Carbonate Rocks 2,000,000
THE EARTH’S SEASONS • The seasonal variations in temperature, and the ratio of day to night hours, are due to the fact that Earth’s axis of rotation is tilted about 23.5° away from perpendicular to the plane of its orbit around the Sun (the ecliptic). • On the first day of summer in the northern hemisphere, the north pole of Earth’s axis is tilted 23.5° toward the Sun, days are longer than nights, and the Sun illuminates the surface at a higher angle from horizontal (both of which contribute to higher average temperatures). • On the first day of winter in the northern hemisphere, the north pole of Earth’s axis is tilted 23.5° away from the Sun, nights are longer than days, and the Sun illuminates the surface at a lower angle from horizontal (both of which contribute to lower average temperatures).
THE EARTH’S SEASONS • The variation of the seasons is opposite in the southern hemisphere than in the northern hemisphere; i.e. it is winter in the southern hemisphere when it is summer in the northern hemisphere, and vice versa. • The first day of spring (vernal equinox) and the first day of fall (autumnal equinox) occur when Earth’s rotation axis is parallel to the Sun, and day and night are of equal length everywhere on Earth. • The slight variations in Earth’s distance from the Sun has very little effect on Earth’s temperature and seasonal variations.
Distribution of Solar Radiation Over Sunlit Hemisphere of Earth
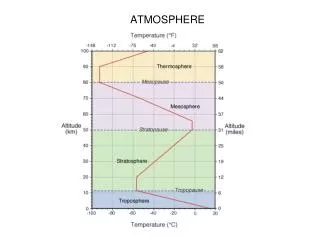
ATMOSPHERIC STRUCTURE • The pressure, temperature, and composition of the atmosphere vary with height above Earth’s surface, as well as with location on Earth’s surface. • In the lower regions of the atmosphere, known as the troposphere, the composition is relatively independent of altitude, and variations are primarily of pressure and temperature, which decrease with altitude. • In the region known as the stratosphere, beginning at about 10 km altitude (the tropopause), pressure continues to decrease with altitude, but temperature increases with altitude. • In the next higher region, known as the mesosphere (middle atmosphere), beginning at about 50 km altitude (the stratopause), temperature again decreases with altitude. • At altitudes greater than about 80 km, known as the mesopause, temperature again increases with altitude, in the region known as the thermosphere. • This region of the atmosphere is also noteworthy in that its composition changes with altitude, with lighter gases becoming more abundant relative to heavier gases (the region known as the heterosphere, as distinct from the lower regions where composition is nearly constant with altitude, known as the homosphere).
ATMOSPHERIC THERMODYNAMICS • The temperature and pressure of the atmosphere vary with altitude in a complex manner, due to a variety of external, as well as internal, influences which can be quantitatively understood using the laws of thermodynamics. • Important aspects include the specific heat of the gas (the amount of energy required to increase the temperature by a given amount), which is different for the cases of constant pressure and of constant volume. • The pressure of a given number, n, of gas atoms or molecules per unit volume is given by: p = nkT, where k is Boltzmann’s constant, and T is the temperature (in degrees Kelvin). • Note also, the specific heat at constant pressure is higher than the specific heat at constant volume, because work has to be done to make the gas expand. • In the case of atmospheric science, the force which compresses the atmosphere, i.e. Earth’s gravity, decreases relatively slowly (as 1/R2, with altitude, where R is distance from Earth’s center).
ATMOSPHERIC SCALE HEIGHT Pressure in a planetary atmosphere decreases with height above the surface, at a rate that depends on the acceleration of gravity, g; the atomic or molecular weight, M, the temperature, T (°K); and Boltzmann’s constant, k: which yields a pressure at altitude h, relative to the pressure p0 at the surface or reference altitude, of where H is the scale height: The scale height is the distance over which the pressure decreases by a factor of 1/e. Note, this relationship is complicated by the fact that temperature, acceleration of gravity g, and, in some cases, average atomic or molecular weight, M, vary with altitude.
THE EARTH’S ATMOSPHERE • Earth’s weather and climate are dependent on the balance between heat input and heat output in the lower atmosphere. • Solar radiation is the primary source of heat energy; however, most of the input to the atmosphere is not directly from the Sun, but is a secondary result of heating Earth’s surface. • Heat transfer as a result of the evaporation and condensation of water is also an important part of atmospheric thermodynamics. • The atmospheric circulation pattern is also influenced by Earth’s rotation, by way of the Coriolis effect. • Weather and climate are also affected by the detailed distribution of land vs. sea surface areas. • The global climate can be influenced by changing concentrations of infrared-trapping “greenhouse gases”, such as CO2 and CH4, in the atmosphere. • Water vapor (H2O) is also an important greenhouse gas, whose concentration in the atmosphere increases with temperature. • Therefore, in principle, increasing amounts of other greenhouse gases could, in principle, cause a catastrophic “run-away greenhouse” event, by increasing the amount of water vapor in the atmosphere.
Typical Temperature Variations with Altitude in Earth’s Atmosphere
Attenuation of Incoming Sunlight by Earth’s Atmosphere Spectral Intensity of Sunlight in Near-Earth Space Spectral Intensity of Sunlight at Earth’s Surface
Attenuation of Incoming Sunlight by Earth’s Atmosphere Incoming Outgoing
METEOROLOGY • The field of atmospheric science which is most well-known and of practical importance to the general public is meteorology, the study of weather. • Meteorology is usually concerned only with the lowest region of the atmosphere, the troposphere. • Weather is influenced not only by vertical, diurnal, and seasonal variations of atmospheric density and temperature, and of solar heating, but also by horizontal variations over Earth’s surface. • Atmospheric winds and circulation are influenced by Earth’s rotation, and by surface conditions (i.e. whether land or sea, topography, and surface temperature). • The advent of weather-monitoring satellites, and of supercomputers, have greatly facilitated the science and application of meteorology in recent years.
Typical Atmospheric Circulation Patterns in the Northern Hemisphere H L
Atmospheric Isobaric Contours and Air Flow Directions 1024 1020 1016 Pressure in Millibars H 1028 1012 1008 1000 L 1004 996
ACTIVE WEATHER PHENOMENA • The topics discussed up to now are typical conditions and motions within Earth’s troposphere. However, as is well known, meteorology also deals with weather conditions which are not typical, but can occur when conditions, such as temperature, humidity, and wind motions, are conducive. In order of increasing strength (but decreasing probability, at any particular location) are the following: • Rainstorms and Snowstorms • Windstorms and Lightning (often associated with rainstorms) • Hurricanes • Tornadoes • Weather conditions of these types depend on the local climate (typical average temperatures, altitudes, and windiness), proximity to oceans or mountains, and time of year. • Although tornadoes can have higher wind velocities than hurricanes, they are much smaller in size and shorter in effective lifetime than are hurricanes.
Viewing Earth’s Atmosphere From Space • One of the most important practical benefits from the U.S. space program has been the development and operational use of space-based observations of Earth’s atmosphere, to detect, measure, and track weather patterns in the troposphere and lower stratosphere. • The benefits of space-based operations include the ability of a satellite, in orbit around Earth, to view (depending on its orbit) the entire Earth, at regular intervals for each location, and/or nearly half of the Earth’s surface at one time, continuously, for long periods of time. • Satellites in low-altitude, polar orbits can view nearly the entire Earth, over long periods of time, but only a small portion of the Earth at one time. • Satellites in high-altitude, equatorial “geosynchronous” orbits can view nearly half of Earth’s surface continuously, over long periods of time, but at lower resolution than achievable with satellites in low-altitude orbits.
Earth Observing Satellites • Satellites in near-polar “sun synchronous” orbits at relatively low altitudes (> 500 km) can view, over a period of about 1 day, nearly the entire Earth with high resolution (but only a small area at any one time). • Satellites in equatorial “geosynchronous” orbits, at about 35,800 km altitude, revolve around Earth once every 24 hours, the same time period that Earth rotates on its axis, and hence appear (to an observer on the Earth) to be always in the same position in the sky (geostationary).
View of Earth and Hurricane Andrew from Geosynchronous Orbit
The Aura Earth Observing System Satellite • The Aura satellite, launched into near-polar Earth orbit July 15, 2004, is the last of the series of Earth Observing System (EOS) research satellites launched by NASA, whose predecessors are the Terra land-observing satellite, and the Aqua water-observing satellite. • The Aura mission is one of scientific research, as distinct from “operational” meteorological satellites, most of which are under the National Oceanic and Atmospheric Administration (NOAA).