Osmosis
Osmosis. Plant Physiology 2009 UNI. Two ways to move water. Bulk flow Osmosis Both move water from high energy to low Differences Source of energy difference What gets moved Structures . Bulk flow. Source of energy difference: Δ pressure Requires open “pipes”

Osmosis
E N D
Presentation Transcript
Osmosis Plant Physiology 2009 UNI
Two ways to move water • Bulk flow • Osmosis • Both move water from high energy to low • Differences • Source of energy difference • What gets moved • Structures
Bulk flow • Source of energy difference: Δpressure • Requires open “pipes” • Everything in the fluid moves at once • Advantages • Move lots of material • Move long distances quickly • Disadvantage • No chance to select what gets moved
Examples of bulk flow Xylem sap is being pulled up. Phloem sap is being pushed down. Heart contraction pushes blood out into vessels.
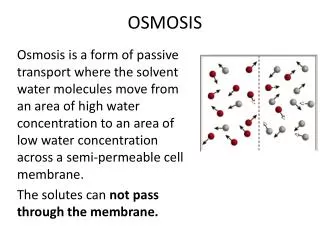
Osmosis • Source of energy difference: • Solutes control energy (potential) of water • Pressure also controls energy (potential) of water • Requires selective membrane between compartments • Lets water through • Does not let solutes through • Examples: water movement between adjacent cells • Restoration of turgor to wilted leaves • Rehydration of cells of marathon runner • Advantage • Lets organism move (just) the water • Disadvantages • Slow • Small volumes moved • Short distances
Lowering water energy with solutes • Start with pure water • Maximum water energy (potential) • Effect of solutes on water energy • Solutes partly tie up water • Solutes reduce water energy • Can water energy by adding solutes • Solute effect on water energy • 0 if no solutes • Negative if solutes present • More solutes? More negative
Seawater pure water Example I of simple osmosis • Water permeable bag allows water to move but not salts • Water moved • From high energy (pure water) • To low energy • Continues until no water left in bag • No pressure differences between bag and surroundings air Seawater air pure water START LATER
fresh water salty water Example II of simple osmosis • Water permeable bag allows water to move but not salts • Water moved • From high water energy (pure water) • To low water energy (water tied up by solutes) • Continues until bag breaks • No pressure differences between bag and surroundings air fresh water air salty water START LATER
Osmotic movement of water • Continues until • energy of water = in both compartments • then molality (not molarity) will be the same in both • or a pressure difference develops • doesn’t happen with flexible bag, animal cells • does happen with stiff cell walls (coming up) • No energy difference = no net movement
Changing water energy with pressure • Start with pure water • Maximum water energy (potential) • Effect of pressure • Can by increasing pressure • Can by lowering pressure • Pressure can be +, 0, - • Pressure-induced differences add to solute-induced differences
Example I of cell osmosis Seawater air Seawater air • Water permeable bag allows water to move but not salts • Water moves • From high energy (pure water) to low energy • Until concentration of solutes (water energy) = inside and out • No pressure differences between bag and surroundings • Cell with membrane shrinks away from wall=plasmolysis typical cell typical cell LATER cell wall START
Example II of cell osmosis • Water permeable bag allows water to move but not salts • Water moved • From high energy (pure water) to low energy • Continues until pressure effect on water energy = solute effect • Big pressure differences between bag and surroundings • Cell volume hardly changes (pressure does) fresh water air fresh water air typical cell typical cell START LATER