Temperature
Temperature Physics 313 Professor Lee Carkner Lecture 2 Thermodynamics Thermodynamics is the study of energy e.g. a cylinder of gas accelerated to high speed has an increased kinetic energy but the same temperature

Temperature
E N D
Presentation Transcript
Temperature Physics 313 Professor Lee Carkner Lecture 2
Thermodynamics • Thermodynamics is the study of energy • e.g. a cylinder of gas accelerated to high speed has an increased kinetic energy but the same temperature • We will discuss the properties of a system and how they relate to internal energy, through both the macroscopic and microscopic points of view
Specify a few basic bulk properties Classical Thermodynamics Specify states of individual units Statistical mechanics Macroscopic Microscopic
Systems • Material separated from its surroundings by a boundary • Closed • e.g. gas in a piston • Open • e.g. an automobile radiator
Processes • State • Process • Quasi - Static (Quasi-Equilibrium) Process • slow changes • Non-quasi static processes are discontinuous
Equilibrium • Define two properties for a system • If the two properties remain constant the system is in equilibrium • A system is in equilibrium with itself if its properties are constant throughout the whole system
System Boundaries • Adiabatic Wall • thermal insulator • Dithermal wall • thermal conductor
Temperature Proxies • Changes in temperature cause changes in other system properties • Two consequences: • We measure T by measuring changes in other properties • If T is not constant, nothing else is • Often given at standard temperature (0 C) or room temperature (20 C)
Thermal Expansion • The degree to which a linear distance varies with temperature is given by: • Change in one dimension (linear expansion) • Very small (~one part in a million per degree) in most cases • Note that a itself is temperature dependant
The Brooklyn Bridge • Built: • Designed: John A Roebling • Chief Engineer: Washington A. Roebling • Height: • Length: • World’s longest suspension bridge until 1903 • To learn more about the Brooklyn Bridge visit your local library: • The Great Bridge : The Epic Story of the Building of the Brooklyn Bridge by David McCullough
Thermal Equilibrium • Two systems separated by a dithermal wall that do not change properties • If the two systems are not in thermal equilibrium they will exchange heat until they are
Zeroth Law • Three systems A, B and T • A and T -- • B and T -- • Then for A and B • Two systems in thermal equilibrium with a third are in thermal equilibrium with each other
Isotherms • Plot pressure and volume • Curve is called an isotherm • Temperature determines thermal equilibrium
Temperature • Take an object T and mark it so that changes in its properties can be measured • Calibrate the measurements so that your scale corresponds to universally accepted situations • e.g. mercury thermometer
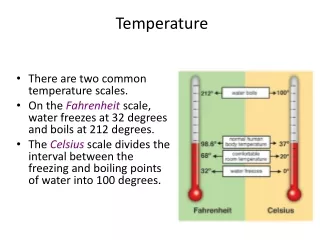
Temperature Scales • Gabriel Daniel Fahrenheit invented the mercury thermometer in 1714 • 32 F is freezing point of water, 212 is chosen for boiling point (32+180) • Anders Celsius introduced his scale in 1742 • William Thomson, Lord Kelvin, developed the absolute (Kelvin) scale from his analysis of ideal engines
Temperatures • Universe (Planck time) ~ • Universe (today) ~ • Average star ~ • Average dust cloud ~ • Planet (Mercury) ~ • Planet (Neptune) ~ • Planet (habitable) ~