Chemical Reactions
120 likes | 249 Views
Learn about reactants, products, catalysts, equations, and types of chemical reactions. Explore balancing equations, net ionic equations, and solubility rules for chemical compounds. Comprehensive guide for beginners.

Chemical Reactions
E N D
Presentation Transcript
By: Indya Evans Chemical Reactions
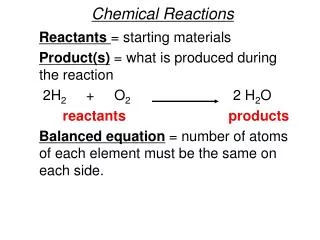
What is a Chemical Reaction? • a process in which one or more substances are changed into others • A change in the arrangement of atoms or molecules to yield substances of different composition and properties • the interaction of two or more chemical substances producing new substances with different properties from the original ones
What is there to know? • Reactants Products • The coefficient is the whole number in front of the elements • Catalyst: is a substance that speeds up a reaction but is not used up in the reaction. • A chemical equation represents the reaction • A balanced equation has an equal number of atoms on both sides of the equation
Symbols • +: used to separate two reactants or two products • : “Yields” separate reactant from product • (s): Solid state after reaction • (l): liquid state after reaction • (g): Gaseous State after reaction • (aq): aqueous State after reaction (meaning the substance is dissolved in water) • heat tells that heat is supplied to the reaction • Pt: indicates the use as a catalyst (for ex: platinum)
Definitions • Law of conservation of mass: mass is conserved • Mass is neither created nor destroyed in a reaction…. That means that one must account for everything! • Law of conservation of charge: same deal except charge…. • Means one must account for all charge within a reaction
The 5 types of Chemical Reactions • Combination Reaction • Equation: R + SRS • Decomposition Reaction • RSR + S • Single- Replacement Reaction • T + RSTS + R • Double- Replacement Reaction • R+ S- + T+ U-R+ U- + T+ S- • The outsides come together + the insides
Combustion reaction 5. Combustion Reaction • Cx Hy + ( x=y/4)O2 xCO2 + (y/2)H2O • Burning fire
How to balance an equation 1st: Identify what type of reaction it is 2nd: list each element and it’s number 3rd: Balance equation by using whole number coefficients EXAMPLE: Li+O2 Li2 O • Decomposition reaction • Li =1 Li=2 O =2 O=1 • 4Li+O22Li2O
Multiplying equation by 2 • When an equation does not have all whole number coefficients to make the equation balanced. The equation is to be multiplied by 2 (only) EXAMPLE: C3H6+O2 CO2+H2O C=3 C=1 H=6 H=2 O=2 O=3 2(C3H6+ 4 1/2 O2 3 CO2+3H2O) • 2 times 4 ½ is 9 which is a whole number! NEW EQUATION • 2C3H6+ 9O26 CO2+ 6H2O • Be sure to multiply the other numbers be 2!
Net Ionic Equations • When writing a complete ionic equation the compound elements split into one element each (and everything is there) • If the ion is unchanged in the equation it can be deleted (spectator ion) • When writing a net ionic equation one only writes what is directly involved in the chemical change
Reminder • KNOW YOUR DIATOMIC ATOMS • H( hydrogen) Ham • N(nitrogen) ~N~ • O (Oxygen) Old • F (Fluorine) Flaming • Cl (Chlorine) Cheese • Br (Bromine) Before • I (Iodine) Ice cream