Atoms
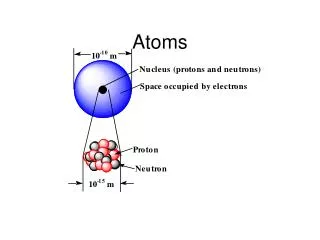
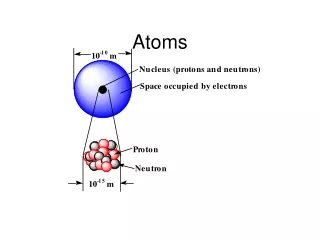
Atoms. Physics Montwood High School R. Casao. More than 100 different elements have been discovered. Each element consists of atoms that contain: a number of protons Z, an equal number of electrons , and a number of neutrons N. The number of protons Z is called the atomic number.

Atoms
E N D
Presentation Transcript
Atoms Physics Montwood High School R. Casao
More than 100 different elements have been discovered. • Each element consists of atoms that contain: • a number of protons Z, • an equal number of electrons , • and a number of neutrons N. • The number of protons Z is called the atomic number. • The lightest atom, hydrogen (H), has Z = 1; helium (He) has Z = 2; lithium (Li) has Z = 3; and so forth. • Nearly all the mass of the atom is concentrated in a tiny nucleus, which contains the protons and neutrons.
The nuclear radius is about 1 to 10 fm (1 fm = 10-5 m). • The distance between the nucleus and the electrons is 0.1 nm; this distance determines the size of the atom. • The chemical and physical properties of an element are determined by the number and arrangement of the electrons in the atom. • Each proton has a positive charge +e so the nucleus has a total positive charge +Z·e. • The electrons are negatively charged (-e), so they are attracted to the nucleus and repelled by each other.
Since electrons and protons have equal but opposite charges and there are an equal number of electrons and protons in the atom, atoms are electrically neutral. • Atoms that lose or gain one or more electrons are electrically charged and are called ions. The Bohr Model • Developed by Niels Bohr to explain the spectra emitted by hydrogen atoms. • By the beginning of the 1900’s, data had been collected on the emission of light by atoms in a gas when they are excited by an electric discharge.
Viewed through a spectroscope with a narrow-slit aperture, the light appears as a discrete set of lines of different colors and wavelengths. • The spacing and intensities of the lines are characteristic of the element. • The wavelengths of these spectral lines could be accurately determined, and much effort went into finding regularities in the spectra. • The line spectra for sunlight, hydrogen, and mercury:
Johann Balmer found that wavelengths of the lines in the visible spectrum of hydrogen can be represented by the formula • Balmer suggested that this might be a special case of a more general expression that would be applicable to the spectra of other elements. • Such an expression was found by Rydberg and Ritz, known as the Rydberg-Ritz formula, gives the reciprocal wavelength as:
Rydberg-Ritz formula: where: • n1 and n2 are integers with n1 > n2 • R is the Rydberg constant, which is the same for all spectral series of the same element and varies only slightly in a regular way from element to element. • For hydrogen, RH = 1.096776 x 107/m. • Rydberg-Ritz formula gives the wavelengths for all the lines in the spectra of hydrogen as well as alkali metals like lithium and sodium.
According to Bohr’s model, the electron of the hydrogen atom moves under the influence of the Coulomb attraction to the positive nucleus according to classical mechanics, which predicts circular or elliptical orbits with the force center at one focus. Energy in a Circular Orbit • For an electron (-e) moving in a circular orbit of radius r about a positive charge Z·e such as the nucleus of a hydrogen atom (Z = 1). • The total energy of the electron can be related to the radius of the orbit.
The potential energy of the electron of charge –e at a distance r from a positive charge Z·e is: • where k is the Coulomb constant. • Kinetic energy K can be expressed as a function of r by using Newton’s second law ΣF = m·a. • Setting the Coulomb attractive force equal to the mass times the centripetal acceleration gives:
Then: • The kinetic energy varies inversely with r like the potential energy. • The magnitude of the potential energy is twice that of the kinetic energy; U = -2·K. • This is a general result in 1/r2 force fields and also holds for circular orbits in a gravitational field. • The total energy is the sum of the kinetic energy and the potential energy:
Mechanical stability is achieved because the Coulomb attractive force provides the centripetal force necessary for the electron to remain in orbit. • Classical electromagnetic theory says that such an atom would be unstable electrically because the electron must accelerate when moving in a circle and therefore radiate electromagnetic energy of frequency equal to its motion. • According to classical theory, such an atom would quickly collapse, the electron spiraling into the nucleus as it radiates away its energy. • Bohr solved the difficulty of the collapsing atom by postulating that only certain orbits, called stationary states, are allowed, and in these orbits the electron does not radiate.
An atom radiates only when the electron makes a transition from one allowed orbit (stationary orbit) to another. • Bohr’s first postulate: nonradiating orbits The electron in the hydrogen atom can move only in certain nonradiating, circular orbits called stationary orbits. • Bohr’s second postulate relates the frequency of radiation to the energies of the stationary states. • If Ei and Ef are the initial and final energies of the atom, the frequency of the emitted radiation during a transition is given by:
Bohr’s second postulate: photon frequency from energy conservation • where h is Planck’s constant • This postulate is equivalent to the assumption of conservation of energy with the emission of a photon of energy h·f • where r1 and r2 are the radii of the initial and final orbits.
To obtain the frequencies implied by the Rydberg-Ritz formula, the radii of stable orbits must be proportional to the squares of integers. • Bohr looked for a quantum condition for the radii of the stable orbits that would yield this result and found he could obtain it if he postulated that the angular momentum of the electron in a stable orbit equals an integer times ħ (h bar; Planck’s constant divided by 2·π).
Bohr’s third postulate: quantized angular momentum where n = 1, 2, … ħ = = 1.055 x 10-34 J·s = 6.582 x 10-16 eV·s • From the Coulomb attractive force equal to the mass times the centripetal acceleration equation, we can solve for v:
Combining the angular momentum equation and the velocity equation:
Solving for the radius of the Bohr atom: • where ao is called the Bohr radius.
Energy Levels • The total energy of the electron in the hydrogen atom is related to the radius of the circular orbit by: • Substituting the quantized values of r given by:
We get: • where n = 1, 2, …
The energies En with Z = 1 are the quantized allowed energies for the hydrogen atom. • Transitions between these allowed energies result in the emission or absorption of a photon whose frequency is given by and whose wavelength is • It is convenient to have the value of h·c in electron-volt·nanometers: h·c = 1240 eV·nm
Since the energies are quantized, the frequencies and wavelengths of the radiation emitted by the hydrogen atom are quantized in agreement with the observed line spectrum. • The figure shows the energy-level diagram for hydrogen. • The energy of the hydrogen atom in the ground state is E1 = 13.6 eV. • As n approaches infinity the energy approaches zero, the highest energy state.
The process of removing an electron from an atom is called ionization, and the energy required to remove the electron is the ionization energy. • The ionization energy of the hydrogen atom, which is the binding energy of the atom, is 13.6 eV. • Transitions from a higher to a lower state are indicated in the figure. • Transitions from n2 = 2 to and n1 = 3, 4, 5, … are known as the Balmer series.
Transitions from n2 = 3 to and n1 = 4, 5, 6, … are known as the Paschen series. • Lyman found the series corresponding to n2 = 1. • Brackett found the series corresponding to n2 = 4. • Pfund found the series corresponding to n2 = 5. • The Balmer series lies in the visible portion of the electromagnetic spectrum.
When electrons are excited, they absorb energy and move to a higher energy level (upward gold arrow). • When they emit light, they move to a lower energy level. • Blue light (blue arrow) is produced when electrons move from level 5 to level 2. • Green light (green arrow) is produced when electrons move from level 4 to level 2. • Red light (red arrow) is produced when electrons move from level 3 to level 2.
Problem example: determine the energy and wavelength of the line with the longest wavelength in the Lyman series. • The Lyman series corresponds to transitions ending at the ground-state energy Ef = E1 = -13.6 eV. • Since λ varies inversely with energy, the transition with the longest wavelength is the transition with the lowest energy, which is that from the first excited state n = 2 to the ground state n = 1.
The energy of the photon is the difference in the energies of the initial and final atomic state: • The wavelength of the photon is: