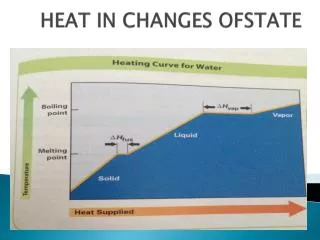
Calculating heat changes
Calculating heat changes. Section 11.4. After reading Section 11.4, you should know:. What Hess’s Law is How to apply Hess’s Law to a chemical equation How to calculate the heat of formation for a given chemical equation. Hess’s Law.

Calculating heat changes
E N D
Presentation Transcript
Calculating heat changes Section 11.4
After reading Section 11.4, you should know: • What Hess’s Law is • How to apply Hess’s Law to a chemical equation • How to calculate the heat of formation for a given chemical equation
Hess’s Law • The heat of 2 reactions added together will equal the heat of a 3rd reaction Adding 2 equations together…you can cancel compounds out IF they are exactly the same on the reactants and products side 2Al + 11/2 O2 --> Al2O3 H = -1669.8 kJ Fe2O3 --> 2Fe + 11/2 O2 H = 824.2 kJ 2Al + Fe2O3 --> 2Fe + Al2O3 H = -844.8 kJ
0 Hf =standard heat of formation Change in enthalpy when 1 mol of new compounds form under standard conditions Table of values on page 316 Ex: Calculate theH0for 2NO(g) + O2 --> 2NO2 Hf NO = 90.37 kJ / mol Hf O2 = 0.0 kJ / mol Hf NO2 = 33.85 kJ / mol 0 0 0
0 1. Find sum ofHf reactants Remember, you have 2 moles of NO 0 2. Find sum ofHf products 3. Find the difference between Hf products and Hf reactants You should have gotten: -113.04 kJ / mol
After reading Section 11.4, you should know: • What Hess’s Law is • How to apply Hess’s Law to a chemical equation • How to calculate the heat of formation for a given chemical equation