Bond types
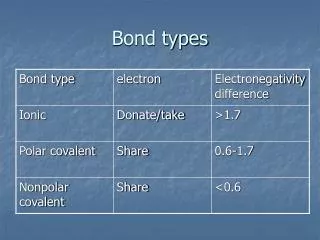
Bond types. Ionic bonds. Bonds are formed by the transfer of electrons. The charges hold the atoms together. Covalent bonds. Elements share valence electrons to form the bond. Chemical reactions. Chemical reactions is the breaking and form of bonds.

Bond types
E N D
Presentation Transcript
Ionic bonds • Bonds are formed by the transfer of electrons. The charges hold the atoms together.
Covalent bonds Elements share valence electrons to form the bond
Chemical reactions • Chemical reactions is the breaking and form of bonds. • More heat: the faster reactions can occur • Exothermic: give off heat • Endothermic absorb heat.
Balancing reactions • The reactants and products must have the same number of atoms. You can not change the subscripts.
Balancing example • Change the coefficient until all atoms are equal
Equilibrium • Where the forward reaction occurs at the same speed as the reverse reaction. All concentrations stay constant.
Fission, Fusion and Radioactive decay Two molecule combine to make a larger Molecule. The Sun Two Hydrogens combine to make Helium A larger molecule splits giving off energy Nuclear energy
Type of Radioactive Particles • Alpha Particle: made of 2 protons and 2 neutrons • Charge = +2 • Mass = 4 • Least amount of energy of radioactive particles, largest radioactive particle • Stopped by a piece of paper (diagram courtesy of the University of Michigan Student Chapter of the Health Physics Society)
Beta Particle: is an emitted electron • Charge = -1 • Mass = .ooo5 • Beta particle is smaller than an alpha particles and move faster. • Beta particles have more penetrating power than alpha particle • Sheet of aluminum foil will stop beta part. (diagram courtesy of the University of Michigan Student Chapter of the Health Physics Society)
Strong force: is one of the strongest forces in nature • The strong force holds protons and neutrons together • Very strong at close distances. Very weak as distance increases • http://www.valdosta.edu/phy/astro/pl_shows/bh_2001/bh/page5.html
Half-Life: the amount of time it takes for a half of a radioactive sample to decay • Hydrogen 3 = 12.3 years • Carbon 14 = 5,730 years • Polonium 211 = .5 seconds • Uranium 235 = 700 million years
Carbon Dating: is used to tell the age of plant and animals (organic compounds) • Scientists look at the ratio of carbon 14 compared to carbon 12 in dead plants and animals • Only useful up to 50,000 years
Measuring Radioactivity • Geiger Counter: is a device that counts charged particles by producing an electric current • Gives off “click” sounds when radioactive particles are detected
Comparison of active and passive solar power. • Passive solar power uses the heat and light from the sun without using any collectors, grids, or cells. • Active solar power systems use focusing mirrors and metal plates to capture the sun’s energy. Solar radiation is absorbed by the collectors and then transfers the heat to air or water. Or photovoltaic cells produce electricity.
Transforming Energy • Law of conservation of Energy: energy is neither created nor destroyed only transformed. • Some energy transformation is not useful for us. • Example: Power lines transform energy into heat. Published January 27, 2011 | By Amanda Kaufmann
Fossil Fuels • Formed by decay of ancient plants and animals • When burned they produce carbon dioxide and water • Petroleum, • Natural Gas, and coal
Petroleum • Fossil Fuel made of Hydrocarbons • Produced by separating the liquid oil in a process called Fractional distillation. • Oil is pumped into the bottom of a tower and heated. Materials rise to the top depending on boiling point and are collected. Some are pulled from the bottom, like asphalt. • Other uses: Plastics, lubricants, asphalt
Natural Gas • Composed mainly of Methane. • Natural gas contains more energy per kilogram than either petroleum or coal. • Produces fewer pollutants than other fossil fuels
Coal • Solid fossil fuel found underground • One fourth of our energy comes from coal. • Coal is formed from organic material deposited in ancient swamps. • Burning coal results in more pollutants
Simple Electric Generator • When the coil is turned, the magnets cause movement in the electrons within the coil. The movement of electrons causes an electric current. magnets coil
Nuclear Power • Energy is released when the nucleus of an atom breaks apart. (Fission) • Nuclear power plants produced 8% of all power in 2003. • There were 104 nuclear reactors at 65 power plants in the US.
16.2 Heat & Thermodynamics • There are 3 ways that thermal energy is transferred from one object to another • Conduction • Convection • Radiation
Conduction: is a heat transfer when 2 objects are touching (direct contact) • There is NO transfer of matter • Conduction works fastest with: • Solids Liquids Gases • The particles of liquids and gases are farther apart than solids, therefore heat is not transferred as quickly
Thermal Conductors: are materials that allow heat and electricity to pass through them easily • Metals • Copper wire for electrical circuits, gold wire in spaceships, pots & pans • Low specific heat values
Thermal insulators: are materials that DO NOT allow heat and electricity to pass through them easily • Non-metals • Wood, plastic, air, glass, fiberglass • Protect items from heat and electricity • High specific heat values
Convection: heat transfer that takes place in fluids (liquids and gases) • HOT AIR/WATER RISES • Convection Currents: currents in liquids or gases, these currents transfer thermal energy (caused by difference in density) • Ex. Pavement, beaker of hot water, ocean currents, weather patterns, wind
Radiation: is the transfer of thermal energy (heat) by waves through space • Radiation happens through air and empty space • Ex. Sun, wood burning stove, fireplace, candles • All objects radiate thermal energy