Alkanes
210 likes | 514 Views
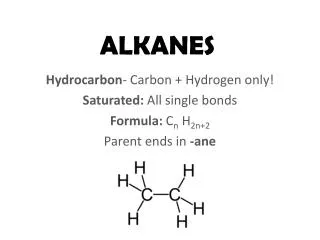
Alkanes. Alkanes are fully saturated hydrocarbons - hydrocarbons have only C’s and H’s - saturated = all single bonds (max. # of H’s) Alkanes have the general formula C n H 2n+2 - example: C 4 H 10 They can be straight-chained or branched

Alkanes
E N D
Presentation Transcript
Alkanes • Alkanes are fully saturated hydrocarbons - hydrocarbons have only C’s and H’s - saturated = all single bonds (max. # of H’s) • Alkanes have the general formula CnH2n+2 - example: C4H10 • They can be straight-chained or branched - example: CH3CH2CH2CH3 vs. (CH3)2CHCH3 • Alkanes, and all other organic compounds, are named according the the IUPAC system (International Union of Pure and Applied Chemistry)
Naming Straight-Chain Alkanes and Alkyl Groups • The names of all alkanes end in -ane • The number of carbons in a straight-chain alkane is indicated by putting a prefix before the -ane • Examples: CH4 = methane CH3-CH3 = ethane CH3-CH2-CH3 = propane • In branched alkanes the substituents (groups attached to the carbon chain) are called alkyl groups • An alkyl group = an alkane with one H removed • Alkyl groups are named by changing the ending of the alkane name to -yl • Example: CH3-CH2- = ethyl
Classification of Carbons in Alkanes • Carbons can be classified by how many other C’s are attached to them: - No C’s = methyl CH4 - 1 C = primary (1) CH3-CH3 - 2 C’s = secondary (2) CH3-CH2-CH3 - 3 C’s = tertiary (3) CH3-CH(CH3)2 - 4 C’s = quaternary (4) C(CH3)4
Naming Branched Alkanes • First find the longest chain of C’s (parent) • Number the C’s in parent chain (begin at end nearest 1rst branch point) • Identify substituents and number by point of attachment to parent chain • Write full name Example: CH3-CH(CH3)-CH2-CH3 = 2-methylbutane • If there is more than one of a substituent a prefix is used and a number is given for each substituent • Example: • CH3-CH2-C(CH3)3 = 2,2-dimethylbutane
Conformations of Alkanes • Because alkanes have only single bonds they have free rotation about those bonds • This allows for different spatial arrangements of the atoms called conformations • Conformations are not the same as isomers • Some conformations are more stable than others, so the compound spends more time in those • For ethane(CH3-CH3): - when all H’s are lined up as you sight down the C-C bond, called eclipsed, this is highest energy - when none of the H’s are lined up, called staggered, this is lowest energy
Haloalkanes • Haloalkanes have one or more halogens replacing hydrogen on an alkane • The halogens are numbered and named as substituents - F = fluoro - Cl = chloro - Br = bromo - I = iodo • If more than one halogen is present, they are named in alphabetical order • Example: CH3-CH(Br)-CH(Cl)-CH3 = 2-bromo-3-chlorobutane
Cycloalkanes • Carbons can also bond together to form rings • Rings with only C’s, H’s and single bonds are called cycloalkanes • Cycloalkanes have the general formula CnH2n • The smallest is cyclopropane (C3H6) - cyclopropane is an unstable molecule - it’s forced to have bond angles of 120between the C’s, while they would normally be 109 (the carbons each have 4 electrons groups and should be tetrahedral) • The only cycloalkanes with little or no strain are cyclopentane (C5H10) and cyclohexane (C6H12) • Most cycloalkanes are not flat because they prefer tetrahedral geometry
Naming Cycloalkanes • Cycloalkanes are named by adding cyclo- to the parent alkane name • Substituents are numbered when there is more than one of them • They are numbered starting at the one that gives the others the lowest numbers • Or, they are numbered alphabetically when there are only two, or both directions are equal
Cis and Trans Isomers • Because cycloalkanes do not have free rotation around the carbons, it matters on which side of the ring a substituent is relative to other substituents • Two substituents on the same side (top or bottom) of the ring are called Cis • Two substituents on opposite sides of the ring are called Trans • Cis and Trans isomers are stereoisomers; they have the same molecular formula, and the atoms are connected in the same order but are arranged in a different spacial orientation
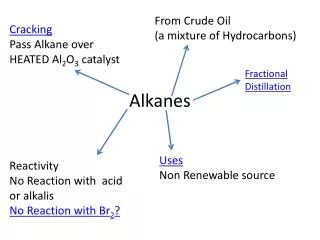
Physical Properties of Alkanes and Cycloalkanes • Alkanes are nonpolar and are not soluble in water • They have low melting and boiling points due to their weak intermolecular forces (dispersion) • They are also less dense than water • Alkanes are mostly obtained from crude oil - the crude oil is fractionated based on boiling pt. - heavier fractions are often “cracked”, put under high heat and pressure, to obtain more gasoline • Alkanes are used extensively as fuels of various types (for heating, cooking, driving, etc.)
Reactions of Alkanes and Cycloalkanes • Because they are nonpolar, and their covalent bonds are strong, alkanes and cycloalkanes are relatively unreactive • The two types of reactions that they do undergo are combustion and halogenation • In combustion reactions, alkanes react with oxygen to form carbon dioxide, water and heat CH4 + 2O2 CO2 + 2H2O + Heat • In halogenation reactions, halogens replace one or more H’s on an alkane (a substitution reaction) CH4 + Cl2 (+ light or heat) CH3Cl + HCl
Mechanism of Alkane Halogenation • These halogenations are radical reactions • Radicals are molecules (or atoms) that have one or more unpaired electrons (the half-headed arrows represent movement of single electrons