Anticoagulant Therapy
410 likes | 1.27k Views
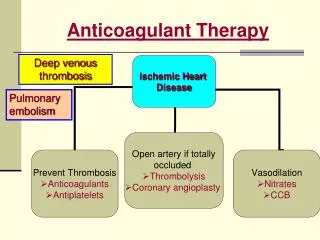
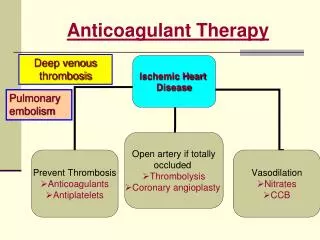
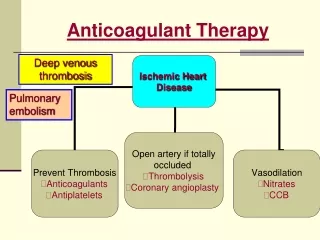
Anticoagulant Therapy. Deep venous thrombosis. Pulmonary embolism. Coagulation Cascade. Antiagoagulant therapy is aimed at: preventing clot in patients at risk Prevent clot extension/ embolisation Deep venous thrombosis (DVT) & pulmonary embolism (PE)

Anticoagulant Therapy
E N D
Presentation Transcript
Anticoagulant Therapy Deep venous thrombosis Pulmonary embolism
Coagulation Cascade • Antiagoagulant therapy is aimed at: • preventing clot in patients at risk • Prevent clot extension/ embolisation • Deep venous thrombosis (DVT) & pulmonary embolism (PE) • Prothrombinase complex comprises the mixture of FVa/FXa in addition to calcium & phospholipid • The presence of phospholipid accelerates thrombin formation by 780-fold
THROMBIN INHIBITORS • Thrombin inhibitors can either inactivate thrombin directly or block thrombin formation • Thrombin can be inhibited irreversibly by glycosaminoglycans like heparin through an antithrombin III-dependent mechanism • The enzyme can be inhibited reversibly by hirudin and hirudin derivatives in an antithrombin III-independent manner • In addition to inhibiting thrombin, the glycosaminoglycans also block thrombin generation
Antithrombin-III Dependent Thrombin Inhibitors Standard Unfractionated Heparin (UFH) • Heparin is a mixture of glycosaminoglycan molecules, which are heterogenous in molecular size • Antithrombin III (ATIII) binding is a necessary requirement for its anticoagulant activity • The mean molecular weight of heparin is 15,000 D
Mode of Action of Heparin • Antithrombin III (ATIII) is a slow progressive inhibitor of thrombin and other clotting enzymes. • Heparin binds to ATIII through a unique pentasaccharide (light blue areas) → conformational change in the reactive center of ATIII → accelerating the rate of ATIII-mediated inactivation of the clotting enzymes • Heparin also promotes the formation of the thrombin-ATIII complex by serving as a template that binds both thrombin and ATIII • ATIII forms a 1:1 irreversible complex with the coagulation enzymes • Once this occurs, the heparin dissociates and can be reused Heparin
To inactivate thrombin, heparin serves as a template and binds both anti-thrombin III (ATIII) and thrombin Binding to ATIII is mediated by the unique penta-saccharide sequence on heparin Binding to thrombin occurs through the heparin-binding domain on the enzyme Conversely, to inactivate factor Xa, heparin needs only to bind to ATIII through its pentasaccharide sequence Heparin inactivates thrombin by binding both ATIII and thrombin Anti-IIa = Anti-Xa activity
Targets for Heparin-ATIII Complex • Heparin/ATIII inactivates several coagulation enzymes including thrombin (factor IIa) and factors Xa, IXa, & XIa • The enzyme most sensitive to inhibition is factor IIa • The next most sensitive enzyme is factor Xa • By inhibiting these two enzymes heparin inhibits both thrombin activity & thrombin formation
Low Molecular Weight Heparins (LMWHs) • Low molecular weight heparins (mean molecular weight 5000 D), prepared by controlled chemical or enzymatic depolymerization of standard unfractionated heparin are about one third the size of starting material • Whereas about one third of the molecules of unfractionated heparin have the unique antithrombin III (ATIII)-binding pentasaccharide, only about 20% of low molecular weight heparin chains contain the pentasaccharide • Enoxaparin, dalteparin & tinzaparin are available LMWHs products
Mechanism of Action of Low Molecular Weight Heparin (LMWH) • All LMWH molecules, which contain the unique pentasaccharide, can catalyze the inactivation of factor Xa by antithrombin III (ATIII) • In contrast, only 25% to 50% of LMWH molecules that have the pentasaccharide sequence also contain at least 13 additional saccharide units to bind to both ATIII & FIIa • As a result, the antithrombin (anti-factor IIa) activity of LMWH is less than its anti-factor Xa activity • Standard heparin has equivalent anti-factor IIa and anti-factor Xa activity because all of the heparin chains that contain the pentasaccharide are long enough to interact with both ATIII & thrombin
Pharmacokinetic Profile of LMWH • LMWH has a more favorable pharmacokinetic profile than standard heparin because LMWH exhibits less binding to plasma proteins & cell surfaces • The reduced binding to plasma proteins results in • Better bioavailability (90% vs. 20% for heparin) • more predictable anticoagulant response • Laboratory monitoring of LMWH activity is not required • Heparin resistance is rare for LMWH • The reduced binding of LMWH to cell surfaces explains why it has a longer half-life than heparin (4 hr vs. 2 hr for heparin), • Given at fixed doses once to twice daily by S.C. route
Biophysical Limitations of Heparin and LMWH • Both heparin and low molecular weight heparin preparations have biophysical limitations because they are unable to inactivate thrombin bound to fibrin, or to subendothelial matrix and to inhibit factor Xa within the prothrombinase complex • Thrombin binds to fibrin where it remains catalytically active • Thrombin bound to fibrin is protected from inactivation by heparin/antithrombin III
Other Injectable Antithrombotic Agents • Fondaparinux, a pentasaccharide,is an AT-III-dependentselective factor Xa inhibitor • It is indicated for the prevention of venous thrombosis associated with orthopedic surgery • Administered >6 hours postoperatively and dose adjusted for renal impairment Tests for Monitoring Antithrombotic Therapy • Prothrombin time (PT)/International Normalization Ratio (INR), usual target is 2-3 times normal • Activated partial thromboplastin time (aPTT)- (serum UFH) • Anti-Xa activityfor LMWHs-treatment in cases of unexpected bleeding & pregnant women
Therapeutic Uses • Heparin should be given either by IV or S.C. injection with onset of action of few minutes and 1-2 hr respectively • LMWHs is given by S.C. route • I.M. injection produces hematoma formation • Treatment of deep-vein thrombosis & pulmonary embolism • Prevention of postoperative venous thrombosis in patients in acute MI phase or one undergoing elective surgery • Reduction of coronary artery thrombosis after thrombolytic treatment • Anticoagulant of choice in pregnant women
Adverse Effects • Bleeding: Bleeding time monitoring is essential. Treatment involves injection of antidote protamine sulphate(1mg Iv for each 100 units of UFH) (reversal of effect) • Thrombosis: AT-III inactivation may lead to potent activation of many clotting factors & hence increasing thrombosis risk • Thrombocytopenia:UFH-induced thrombocytopenia (HIT) is a life-threatening immune reaction that occurs in up to 3% of patients on heparin therapy for 5-14 days • It induces platelet activation & endothelial damage with enhanced thrombi formation & paradoxical thrombosis • A non-immunologic reversible HIT may occur in early phase of therapy due to direct effect of UFH on platelets • LMWHs, though of lower risk, are contraindicated with HIT
Adverse Effects • Osteoporosis occurs with large doses of UFH >20,000 U/day for 6 months or longer • Hyperkalemia rarely occurs with UFH • It is attributed to inhibition of aldostetone secretion • It occurs with both low- & high-dose UFH therapy • Onset is quick within a week after therapy initiation • It is reversible by therapy discontinuation • Diabetic & renal failure patients are at higher risk • Hypersensitivity:(Antigenicity due to animal source) • rarely occurring reactions include urticaria, rash, rhinitis, angioedema & reversible alopecia
Contraindications • Hypersensitivity to heparin • Active bleeding or hemophilia • Significant throbocytopenia, purpura • Severe hypertension • Intracranial hemrrhage • Ulcerative GIT lesions • Active TB • Recent surgery in CNS, eye • Advanced hepatic or renal disease
Direct Thrombin InhibitorsHirudin Hirugen & Hirlug A, Hirudin • A leech-derived protein, a potent & specific inhibitor of thrombin • It binds to both the substrate recognition site and the catalytic center. The hirudin-thrombin complex slowly dissociates B, Hirugen • A synthetic peptide analogue of the carboxy terminal of hirudin • It binds to the substrate recognition site of thrombin
Direct Thrombin Inhibitors (DTI) • C, Hirulog is a synthetic bivalent inhibitor of thrombin comprised of a catalytic site inhibitor linked to hirugen. Thus, hirulog interacts with both the substrate recognition site and the catalytic center of thrombin. • D, Catalytic site inhibitors interact with the active center of thrombin
Inhibition of Bound Thrombin • Neither heparin/ATIII nor LMWH/ATIII are an effective inhibitor of fibrin-bound thrombin because the heparin-binding site on thrombin is masked when the enzyme is bound to fibrin • In contrast, the ATIII-independent thrombin inhibitors are able to inactivate fibrin-bound thrombin as well as free thrombin
In vivo studies with direct thrombin inhibitors • In experimental animals, hirudin, hirulog, and inhibitors of the catalytic site of thrombin are more effective than heparin in preventing extension of venous thrombosis, preventing platelet-dependent arterial thrombosis, and accelerating thrombolysis • Preliminary studies in humans also suggest that the direct thrombin inhibitors are more effective than heparin in venous thrombosis, in unstable angina, and in the setting of thrombolytic therapy
Clinically Approved Direct Thrombin Inhibitors • Lepirudin, recombinant hirudin-like peptide, has been approved for IV anticoagulant use in HIT patients, has renal clearance • It has potential use in unstable angina patients (Circulation 2001; 103: 1479) • Bivaluridin, a bivalent DTI, used by IV route for patients undergoing percutaneous coronary intervention • Argatroban, a small monovalent (thrombin active site only) molecule, with DTI activity, used similarly in HIT patients, has hepatic clearance • aPTT is used to monitor activity for these agents
DIRECT FACTOR Xa INHIBITORS • There are two direct factor Xa inhibitors, the tick anticoagulant peptide (TAP), originally isolated from the soft tick Ornithodoros moubata and antistasin, derived from the Mexican leech • Both inhibitors are now available by recombinant technology • Studies in animals indicate that both TAP and antistasin are effective antithrombotic agents in experimental models of arterial thrombosis
DIRECT FACTOR Xa INHIBITORS • Differ from heparin and low molecular weight heparins in two ways: 1) they inactivate factor Xa independent of antithrombin III (ATIII); and 2) in addition to inactivating free factor Xa, there is evidence that these agents also are able to inactivate factor Xa within the prothrombinase complex
Oral Anticoagulants Vitamin K Antagonists (The Coumarins) • Vitamin K is crucial co-factor for the hepatic synthesis of clotting factors II, VII, IX & X • Vitamin K catalyses the ɣ-carboxylation of glutamic acid residues in the mentioned factors via a vitamin K-dependent carboxylase • The ɣ-carboxyglutamyl residues bind Ca2+ to enable interaction with phosphlipids
The reduced vit K is converted into vitamin K epoxide which is reduced back by vitamin K reductasethe target enzyme which warfarin inhibits This results in the production of inactive clotting factors lacking ɣ-carboxyglutamyl residues Vitamin K AntagonistsWarfarin
Vitamin K AntagonistsWarfarin • Onset:Effect of a single dose starts only after 12-16 hrs (unlike heparin) & lasts for 4-5 days although its quick GIT absorption • Clinical anticoagulant activity needs several days to develop (four half-lives of clotting factors needed to elapse before steady state) • This may be related to the elimination half-lives of the concerned clotting factors (6-72 hrs) (Factor II: 40-72 hrs, X<48hrs) • Overlap heparin & warfarin therapy to overcome delayed warfarin activity & warfarin-inhibition of the anticoagulant protein C & S
Vitamin K AntagonistsWarfarin • Warfarin has 100% oral bioavailability, powerful plasma protein binding & long plasma t1/2 of 36 hrs • A loading high dose followed by maintenance dose is adjusted • Warfarin is contraindicated with pregnancy as it crosses the placental barrier and is teratogenic in the first trimester & and induce intracranial hemorrhage in the baby during delivery • Warfarin is metabolized by hepatic Cytochrome P450 enzymes with half-life of 40 hrs
Potentiating warfarin Inhibitors of hepatic P450 enzymes (cimetidine, co-trimoxazole, imipramine) Platelet aggregation inhibitors (NSAIDs, aspirin) 3rd G cephalosporins Drugs displacing warfarin from binding sites (NSAIDs) Drugs reducing the availability of vitamin K Hepatic disease & hyperthyroidism Inhibiting Warfarin Vitamin K in some parenteral feed Inducers of hepatic P450 enzymes (rifampicin, barbiturates, … etc) Reduction of GIT absorption (colestyramine) Diuretics Hypothyroidism Warfarin Drug Pharmacokinetic & Pharmacodynamic Interactions
Warfarin Side-Effects • Drug-drug interactions • Bleeding disorder; monitor anticoagulant effect by measuring PT or INR, reversal of action: • Minor bleeding: stop therapy + oral Vitamin K • Severe Bleeding: stop therapy + I.V. Vitamin K • Fresh-frozen plasma, recombinant factor VIIa or prothrombin complex may be used