Immunization
E N D
Presentation Transcript
Immunization Dr. Tanveer Ashraf
Objectives • Types of vaccination • Contraindications • Discussion of individual vaccines • Preparations • Administration • Precautions and contraindications • Side effects
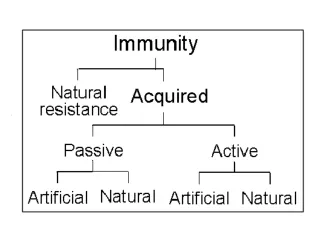
Active Immunization • Types • Live attenuated • Virus Measles, mumps, rubella • Bacteria BCG • Killed • Virus Hepatitis B • Bacteria • Whole Pertussis • Toxoid Tetanus • Polysaccharide Meningoccocal
Human Immune Serum Globulin • Specific • IM Hepatitis B (HBIG) • Rabies (RIG) • Tetanus (TIG) • Varicella (VZIG) • IV CMV (CMV-IG) • RSV (RSV-IG)
Human Immune Serum Globulin • Non-specific • IM Immune serum globulin(ISG) • Hepatitis A • Measles • IV Intravenous immune globulin(IGIV) • Antibody deficiency disorders • ITP • Kawasaki syndrome
Passive Immunization (Cont) • SPECIFIC EQUINE ANTIBODIES (IM) • BOTULISM ANTITOXIN • DIPHTERIA ANTITOXIN • TETANUS ANTITOXIN • SNAKE & SPIDER ANTI-VENOM • MONOCLONAL ANTIBODIES (IV) • ANTI-ENDOTOXIN ANTIBODIES
RECOMMENDED AGE VACCINE(S) BIRTH BCG, Hepatitis B (HBV) 2 MONTHS [DTP, HIB, HBV] Injectable Polio Vaccine (IPV) 4 MONTHS [DTP, HIB, HBV] Oral Polio Vaccine (OPV) 6 MONTHS [DTP, HIB, HBV] Oral Polio Vaccine (OPV) 9 MONTHS Measles vaccine (mono) 12 MONTHS Oral Polio Vaccine (OPV) MMR Varicella 1 8 MONTHS Oral Polio Vaccine (OPV) DTP, HIB Hepatitis A 24 MONTHS Hepatitis A 4 – 6 YEARS Oral Polio Vaccine (OPV) DTP MMR Varicella
General Contraindications • Moderate or severe illness with or without fever • Anaphylactic reaction to vaccine or vaccine constituent • Live attenuated vaccines • Pregnant women • Immunocompromised / Immunosuppressed children • within 3-11 months of immunoglobulin administration
Invalid Contraindications • Mild to moderate local reaction • Mild acute illness with or without low grade fever • Current antimicrobial therapy • Convalescent phase of illnesses • Prematurity and low birth weight • History of penicillin or other nonspecific allergies • Malnutrition
Immunization Of Special Groups • IMMUNOCOMPROMISED HOSTS • Avoid MMR, measles (may be used in HIV) • Avoid OPV; use IPV for these children and their household contacts • PRETERM INFANTS • Treat as term babies • Avoid OPV in hospital • Influenza vaccine in BPD • may delay HBV if <2 kg & mother is HBsAG negative
Bacillus Calmette‑Guerin Vaccine (BCG) • INDICATIONS • All newborns • All tuberculin negative infants • PRECAUTIONS & CONTRAINDICATIONS(CI): • Give only to PPD negative children • CI in persons with immunodeficiencies • CI during pregnancy
Bacillus Calmette‑Guerin Vaccine (BCG) • SIDE EFFECTS • Local • Skin ulceration, regional lymphadenitis • Subcutaneous abscess • Generalized • Anaphylaxis, generalized BCG infection • (rare): osteitis
Hepatitis B • PREPARATIONS • ENGERIX‑B • RECOMBIVAX HB
Hepatitis B • ADMINISTRATION: • 0.5 ‑1 ML, anterolateral thigh or deltoid • IM injection • at 0,1 and 6 months • SIDE EFFECTS: • Pain • Arthralgia
Poliomyelitis Prophylaxis • PREPARATIONS • (A) Oral (OPV) SABIN (Live attenuated) • (B) Inactivated (eIPV) SALK (killed) • ADMINISTRATION • OPV 2 drops orally • eIPV SC injection • PRECAUTIONS & CONTRAINDICATIONS(CI) • (a)OPV pregnancy, immunodeficiency • (b)IPV neomycin hypersensitivity • ADVERSE REACTIONS • OPV paralytic disease (rare)
Diphtheria, Tetanus & Pertussis (DTP) • PREPARATIONS • < 7 years : DTP, DT, DTaP (acellular pertussis vaccine) • > 7 years : Td, TdaP • ADMINISTRATION • IM
Diphtheria, Tetanus & Pertussis (DTP) • CONTRAINDICATIONS (CI) • Encephalopathy within 7 days • Progressive or unstable neurological disorders • Anaphylactic reaction to a previous dose • PRECAUTIONS • severe systemic reactions such as • Temp > 40.50C • persistent inconsolable crying > 3 hours • Collapse episodes • Convulsions
Measles, Mumps & Rubella (MMR) • PREPARATIONS: • MEASLES. • MMR. • ADMINISTRATION: • SC. • INDICATIONS: • Primary immunization at 1 & 6 years.
Measles, Mumps & Rubella (MMR) • PRECAUTIONS & CONTRAINDICATIONS • Pregnancy • Anaphylaxis to eggs • Immunodeficiency and immunosuppression • Immunoglobulins within 3-11 months
Measles, Mumps & Rubella (MMR) • ADVERSE REACTIONS • Fever ,rash (7 days post vaccination) • Arthralgia , arthritis (rubella) • Encephalitis [rare] (measles, mumps) • Suppression of PPD skin test (measles) • Convulsions in prone children(measles) • Thrombocytopenia
Haemophilus Influenzae Type B • ADMINISTRATION • IM • 2,4,6 months, booster at 15 months • INDICATIONS • Children under 5 years of age • High risk children • SIDE EFFECTS: • Local pain and erythema • Mild fever
Varicella Prophylaxis • PREPARATIONS: • Varivax vaccine (MSD) • ADMINISTRATION: • 0.5 ml IM • 12 months and above……..2 doses
Varicella Prophylaxis • INDICATIONS: • All children 12 months‑18 years: (if no history of varicella) • EFFICACY: • 70‑90%
Varicella Prophylaxis • PRECAUTIONS & CONTRAITNDICATIONS: • Immunocompromised patients • Within 5 months of IG • Children on long term salicylates • SIDE EFFECTS: • Fever , rash • Zoster
Hepatitis A • NATURE OF VACCINE: • Killed formalin inactivated vaccine. • PREPARATIONS: • Various preparations available • e.g. Havrix 720 Junior, Havrix 1440 etc.
Hepatitis A • INDICATIONS: • children 1 year and above • Susceptible children in endemic areas • Chronic liver disease • Hemophilia
Hepatitis A • ADMINISTRATION: • IM injection • 2 doses, at least 6 months apart • ADVERSE REACTIONS: • Local reactions, fever • Rare: anaphylaxis
Pneumococcal Prophylaxis • PREPARATIONS: • Purified capsular polysaccharide of 23 serotypes of Streptococcus pneumoniae • 7 valent conjugated vaccine • ADMINISTRATION: • IM / SC • 1 dose/booster 5 years
Pneumococcal Prophylaxis • INDICATIONS: • Primary vaccination (conjugate vaccine) • children 2 yr. or older with • Anatomical or functional asplenia • Sickle cell disease • Nephrotic syndrome • Immunosuppression
Pneumococcal Prophylaxis • SIDE EFFECTS: • Soreness , erythema, fever, myalgia • Anaphylactic reactions (rare)
Meningococcal Prophylaxis • PREPARATIONS: • monovalent (A or C) • bivalent (A & C) • quadrivalent (A,C,Y & W‑135) • quadrivalent conjugate quadrivalent • ADMINISTRATION: • SC
Meningococcal Prophylaxis • INDICATIONS: • Control of outbreaks • Children with complement deficiencies or asplenia • SIDE EFFECTS: • local erythema and discomfort • transient fever
Influenza Virus • Nature of vaccine: • Killed vaccine. • Live attenuated • Preparations: • whole and “split virus” vaccines. • “split virus” vaccines are recommended for children 6 months and older. • composition of the vaccine is changed annually.
Influenza Virus • ADMINISTRATION: • IM (killed). • Live attenuated (intranasal). • 1 dose during influenzae season. • Children 6months-9 years should receive an additional dose, 4 weeks after the 1st dose, if not previously immunized.
Influenza Virus • Indications: • chronic cardio-respiratory disease • asthma • cystic fibrosis • bronchopulmonary dysplasia
Influenza Virus • Indications: • Sickle cell anemia. • Chronic salicylate therapy. • Diabetes mellitus. • Chronic renal disease. • Chronic metabolic disease. • immunosuppressive conditions: cancer, HIV etc. • Hospital personnel with significant patient contact.
Influenza Virus • Contraindication: • Anaphylaxis to previous dose. • Hypersensitivity to eggs. • Adverse Reaction: • Soreness at injection site. • Allergic response. • Guillain-Barré Syndrome.
New Vaccines • Rota virus vaccines • Human papilloma virus vaccine